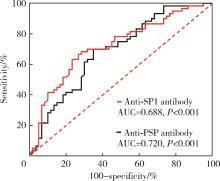
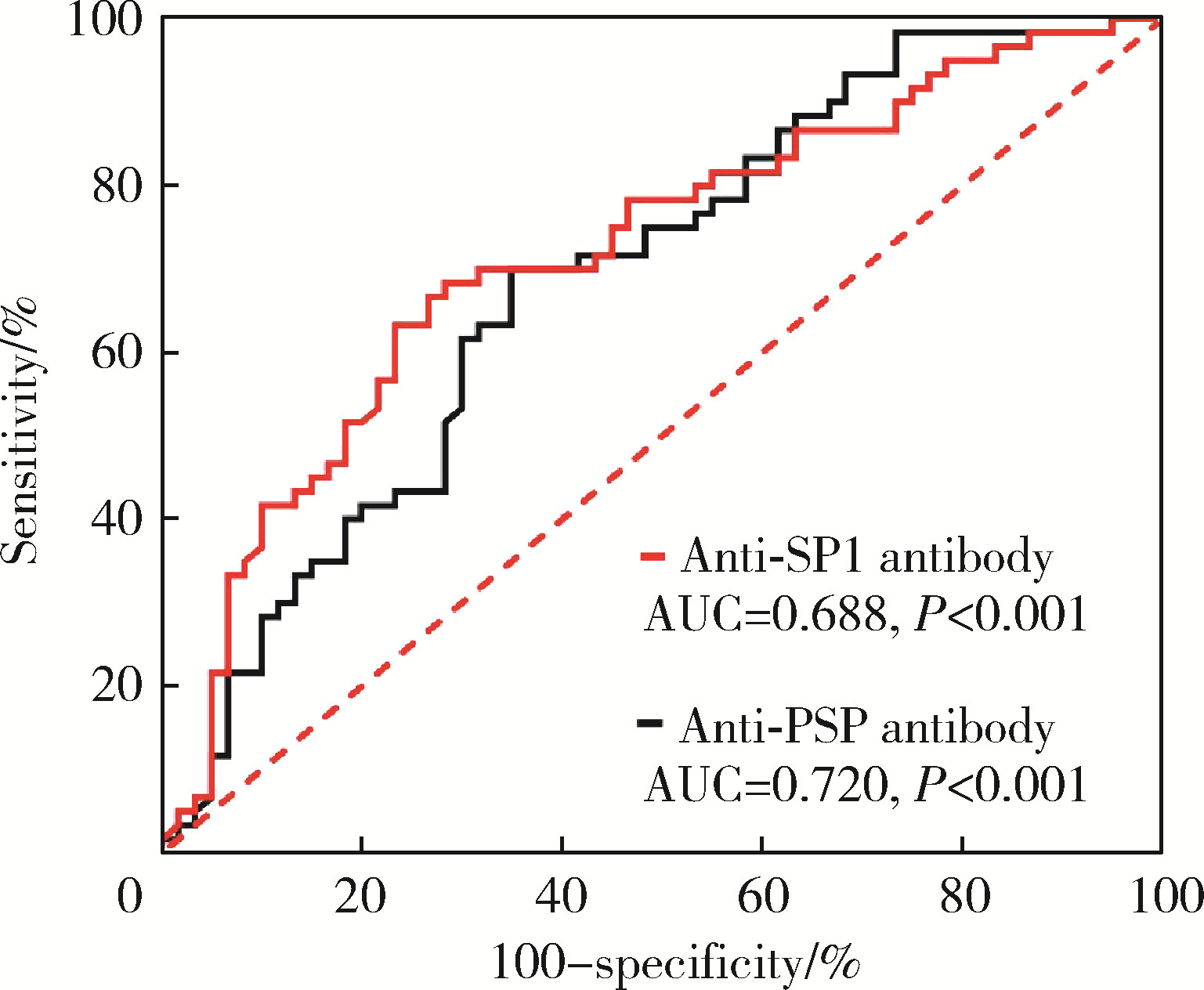
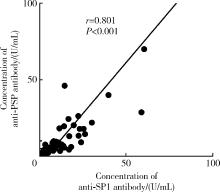
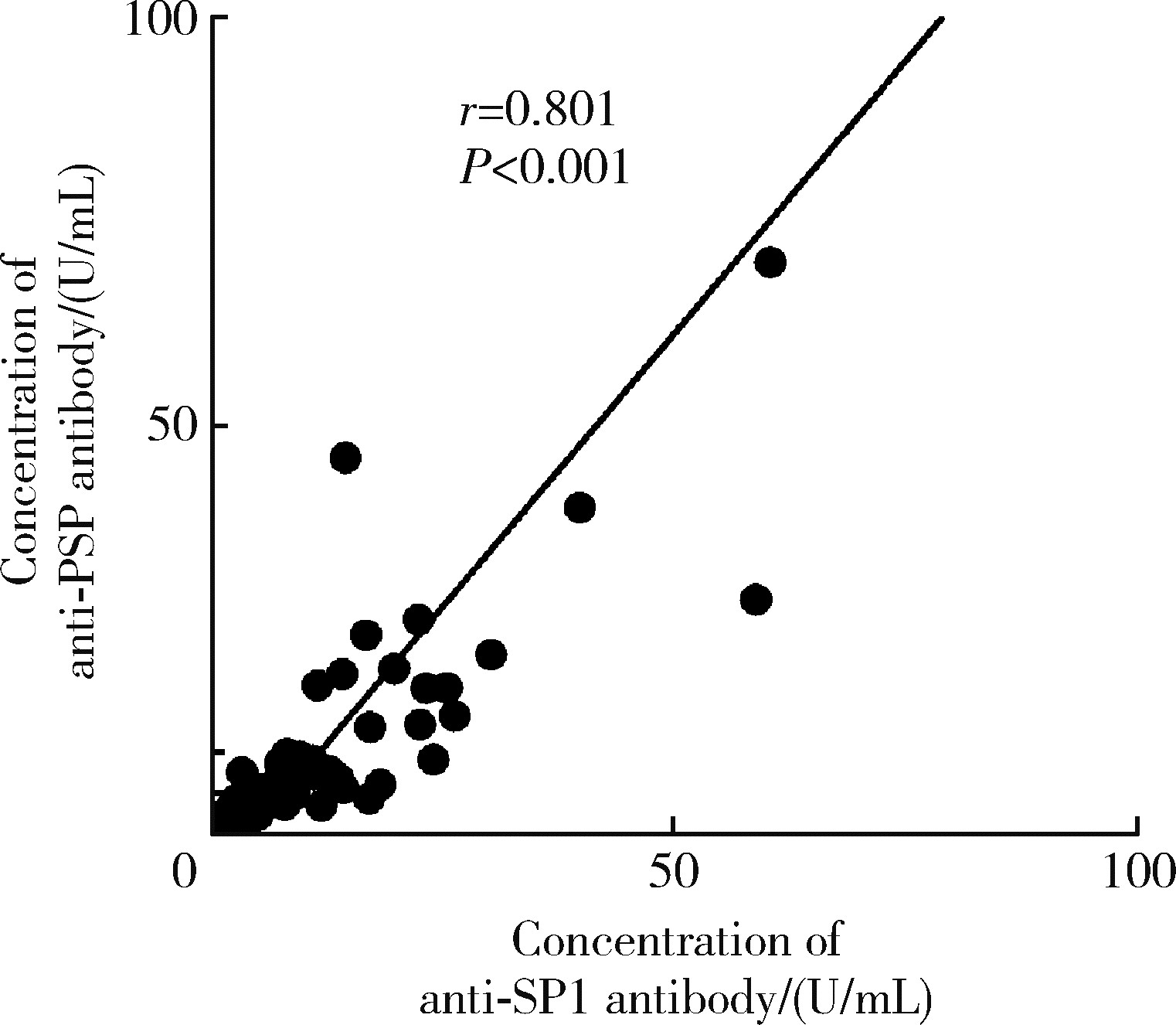
Journal of Peking University (Health Sciences) ›› 2024, Vol. 56 ›› Issue (5): 845-852. doi: 10.19723/j.issn.1671-167X.2024.05.015
Previous Articles Next Articles
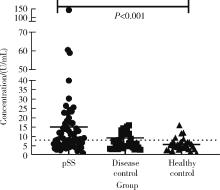
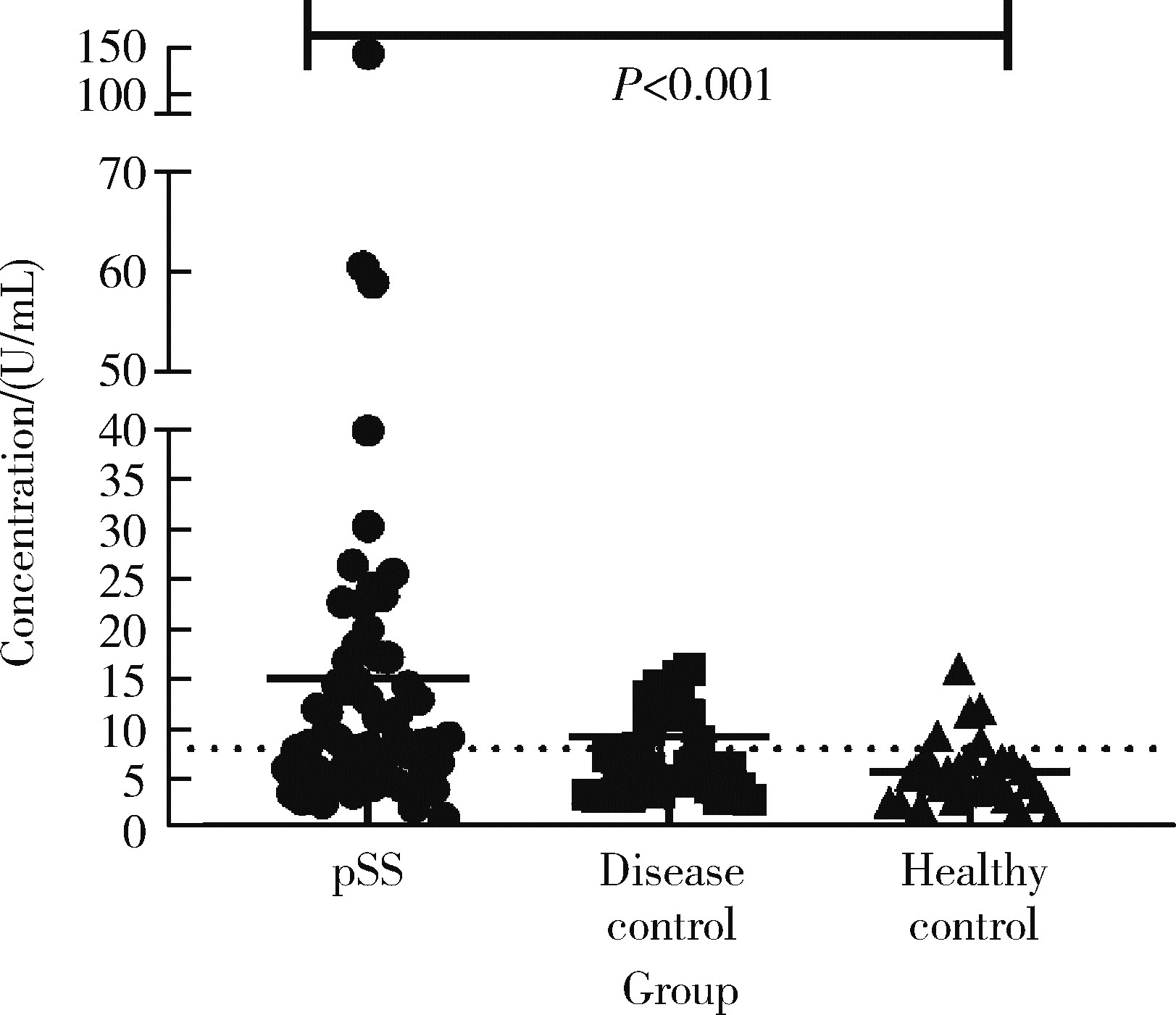
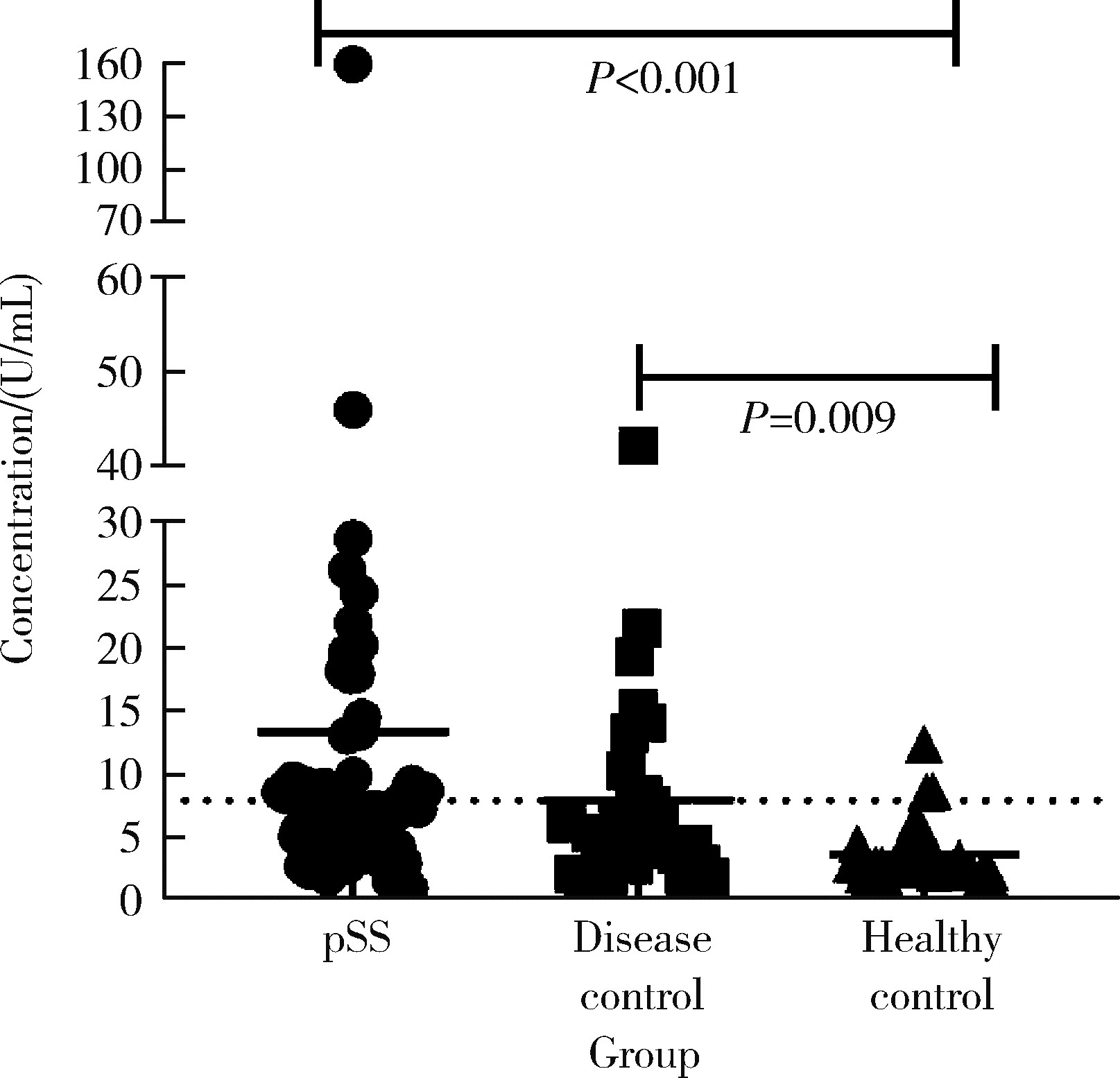
Diagnostic values of anti-salivary gland protein-1 antibody combined with anti-parotid secretory protein antibody for Sjögren's syndrome
Yushu YANG, Xuan QI, Meng DING, Wei WANG, Huifang GUO, Lixia GAO*( )
)
- Department of Rheumatology and Immunology, the Second Hospital of Hebei Medical University, Shijiazhuang 050011, China
CLC Number:
- R593.2
| 1 | Goules AV , Argyropoulou OD , Pezoulas VC , et al. Primary Sjögren's syndrome of early and late onset: Distinct clinical phenotypes and lymphoma development[J]. Front Immunol, 2020, 11, 594096. |
| 2 | Vitali C , Bombardieri S , Jonsson R , et al. Classification criteria for Sjögren's syndrome: A revised version of the European criteria proposed by the American-European Consensus Group[J]. Ann Rheum Dis, 2002, 61 (6): 554- 558. |
| 3 | Shiboski SC , Shiboski CH , Criswell L , et al. American College of Rheumatology classification criteria for Sjögren's syndrome: A data-driven, expert consensus approach in the Sjögren's International Collaborative Clinical Alliance cohort[J]. Arthritis Care Res (Hoboken), 2012, 64 (4): 475- 487. |
| 4 | Shiboski CH , Shiboski SC , Seror R , et al. 2016 American College of Rheumatology/European League Against Rheumatism classification criteria for primary Sjögren's syndrome: A consensus and data-driven methodology involving three international patient cohorts[J]. Ann Rheum Dis, 2017, 76 (1): 9- 16. |
| 5 | Hoshina Y , Wong KH , Galli J , et al. Neurologic involvement in seronegative primary Sjögren's syndrome with positive minor salivary gland biopsy: A single-center experience[J]. Front Neurol, 2023, 14, 1174116. |
| 6 | Chen J , He Q , Yang J , et al. Anti-SSA/SSB-negative primary Sjögren's syndrome showing different clinical phenotypes: A retrospective study of 934 cases[J]. Adv Rheumatol, 2023, 63 (1): 21. |
| 7 | Bodeutsch C , de Wilde PC , Kater L , et al. Labial salivary gland biopsy in Sjögren's syndrome[J]. Neth J Med, 1992, 40 (3/4): 148- 157. |
| 8 | Theander E , Jonsson R , Sjöström B , et al. Prediction of Sjögren's syndrome years before diagnosis and identification of patients with early onset and severe disease course by autoantibody profiling[J]. Arthritis Rheumatol, 2015, 67 (9): 2427- 2436. |
| 9 | Jonsson R , Theander E , Sjöström B , et al. Autoantibodies present before symptom onset in primary Sjögren syndrome[J]. JAMA, 2013, 310 (17): 1854- 1855. |
| 10 | Lee AYS , Lin MW . Serological intermolecular epitope spreading in a patient with primary Sjögren's syndrome[J]. BMJ Case Rep, 2023, 16 (5): e254632. |
| 11 | Mona M , Mondello S , Hyon JY , et al. Clinical usefulness of anti-muscarinic type 3 receptor autoantibodies in patients with primary Sjögren's syndrome[J]. Clin Exp Rheumatol, 2021, 39 (4): 795- 803. |
| 12 | Alam J , Koh JH , Kwok SK , et al. Functional epitopes for anti-aquaporin 5 antibodies in Sjögren syndrome[J]. J Dent Res, 2017, 96 (12): 1414- 1421. |
| 13 | Alam J , Koh JH , Kim N , et al. Detection of autoantibodies against aquaporin-5 in the sera of patients with primary Sjögren's syndrome[J]. Immunol Res, 2016, 64 (4): 848- 856. |
| 14 | Tjensvoll AB , Lauvsnes MB , Zetterberg H , et al. Neurofilament light is a biomarker of brain involvement in lupus and primary Sjögren's syndrome[J]. Journal of Neurology, 2021, 268 (4): 1385- 1394. |
| 15 | Shen L , Gao C , Suresh L , et al. Central role for marginal zone B cells in an animal model of Sjögren's syndrome[J]. Clin Immunol, 2016, 168, 30- 36. |
| 16 | Jin Y , Li J , Chen J , et al. Tissue-specific autoantibodies improve diagnosis of primary Sjögren's syndrome in the early stage and indicate localized salivary injury[J]. J Immunol Res, 2019, 2019, 3642937. |
| 17 | Shen L , Suresh L , Lindemann M , et al. Novel autoantibodies in Sjögren's syndrome[J]. Clin Immunol, 2012, 145 (3): 251- 255. |
| 18 | Xuan J , Wang Y , Xiong Y , et al. Investigation of autoantibodies to SP-1 in chinese patients with primary Sjögren's syndrome[J]. Clin Immunol, 2018, 188, 58- 63. |
| 19 | Xian Z , Fu D , Liu S , et al. Association between B cell growth factors and primary Sjögren's syndrome-related autoantibodies in patients with non-Hodgkin's lymphoma[J]. J Immunol Res, 2019, 2019, 7627384. |
| 20 | Bunya VY , Massaro-Giordano M , Vivino FB , et al. Prevalence of novel candidate Sjögren syndrome autoantibodies in the penn Sjögren's International Collaborative Clinical Alliance Cohort[J]. Cornea, 2019, 38 (12): 1500- 1505. |
| 21 | Shen L , Kapsogeorgou EK , Yu M , et al. Evaluation of salivary gland protein 1 antibodies in patients with primary and secondary Sjögren's syndrome[J]. Clin Immunol, 2014, 155 (1): 42- 46. |
| 22 | Applbaum E , Lichtbroun A . Novel Sjögren's autoantibodies found in fibromyalgia patients with sicca and/or xerostomia[J]. Autoimmun Rev, 2019, 18 (2): 199- 202. |
| 23 | de Langhe E , Bossuyt X , Shen L , et al. Evaluation of autoantibodies in patients with primary and secondary Sjögren's syndrome[J]. Open Rheumatol J, 2017, 11, 10- 15. |
| 24 | Hubschman S , Rojas M , Kalavar M , et al. Association between early Sjögren markers and symptoms and signs of dry eye[J]. Cornea, 2020, 39 (3): 311- 315. |
| 25 | Geetha C , Venkatesh SG , Dunn BH , et al. Expression and anti-bacterial activity of human parotid secretory protein (PSP)[J]. Biochem Soc Trans, 2003, 31 (Pt 4): 815- 818. |
| 26 | Karakus S , Baer AN , Akpek EK . Clinical correlations of novel autoantibodies in patients with dry eye[J]. J Immunol Res, 2019, 2019, 7935451. |
| 27 | Burbelo PD , Ferré E , Chaturvedi A , et al. Profiling autoanti-bodies against salivary proteins in sicca conditions[J]. J Dent Res, 2019, 98 (7): 772- 778. |
| 28 | Thatayatikom A , Jun I , Bhattacharyya I , et al. The diagnostic performance of early Sjögren's syndrome autoantibodies in juvenile Sjögren's syndrome: The university of florida pediatric cohort study[J]. Front Immunol, 2021, 12, 704193. |
| 29 | Lee KA , Kim KW , Kim BM , et al. Clinical and diagnostic signi-ficance of serum immunoglobulin A rheumatoid factor in primary Sjögren's syndrome[J]. Clin Oral Investig, 2019, 23 (3): 1415- 1423. |
| 30 | Zhong H , Wang Y , Yang P , et al. Hyperglobulinemia predicts increased risk of mortality in primary Sjögren's syndrome: Based on a Chinese multicentre registry[J]. Mod Rheumatol, 2023, 34 (1): 137- 143. |
| 31 | Kota SK , Pernicone E , Leaf DE , et al. BPI fold-containing family a member 2/parotid secretory protein is an early biomarker of AKI[J]. J Am Soc Nephrol, 2017, 28 (12): 3473- 3478. |
| [1] | Hui WEI, Jingfeng ZHANG, Zhongqiang YAO, Jinxia ZHAO. Clinical characteristics and relevant factors of rheumatoid arthritis patients with anemia of chronic disease [J]. Journal of Peking University (Health Sciences), 2026, 58(2): 307-312. |
| [2] | Jiyan CHU, Ping LI, Jing TIAN, Diyu FU, Lin GUO, Rui SUN, Yadi LI. Expression of the melanoma 2-mediated pyroptosis pathway in peripheral blood mononuclear cells of patients with idiopathic inflammatory myopathies [J]. Journal of Peking University (Health Sciences), 2026, 58(1): 175-183. |
| [3] | Yuan LIU, Guixiu SHI. Change from Sjögren syndrome to Sjögren disease [J]. Journal of Peking University (Health Sciences), 2025, 57(6): 1015-1017. |
| [4] | Yirui LIAN, Jingxuan LIU, Liang ZHAO, Jing ZHAO, Sitian ZANG, Yuhui LI. Rheumatic disease spectrum and immunological profile of anti-PM/Scl antibodies in idiopathic inflammatory myopathies [J]. Journal of Peking University (Health Sciences), 2025, 57(6): 1018-1023. |
| [5] | Tao WU, Jianzi LIN, Yafeng ZHU, Jianda MA, Peiwen JIA, Lijuan YANG, jie PAN, Yaowei ZOU, Ying YANG, Ye LU, Lie DAI. Serum inter-alpha-trypsin inhibitor heavy chain H3 is identified as a potential biomarker for myopenia in patients with rheumatoid arthritis using proteomic profiling [J]. Journal of Peking University (Health Sciences), 2025, 57(6): 1024-1031. |
| [6] | Wenhao LIN, Yang XIE, Fangqing WANG, Shuying WANG, Xiangjun LIU, Fanlei HU, Yuan JIA. Single-cell RNA sequencing of B cells reveals molecular typing in Sjögren syndrome [J]. Journal of Peking University (Health Sciences), 2025, 57(6): 1032-1041. |
| [7] | Zhao XIANG, Li YANG, Jing YANG. Untargeted metabolomics reveals differential serum metabolites and metabolic pathways in patients with primary Sjögren syndrome and thrombocytopenia [J]. Journal of Peking University (Health Sciences), 2025, 57(6): 1042-1050. |
| [8] | Yayun ZHAO, Mengfan NI, Xue LI, Bei WANG, Gong CHENG, Jing HE, Yuebo JIN. Clinical efficacy and safety of rituximab in treating renal injury in primary Sjögren syndrome [J]. Journal of Peking University (Health Sciences), 2025, 57(6): 1051-1060. |
| [9] | Jiajing PENG, Li CUI. Clinical features and prognosis of sarcoidosis with ocular lesions as the initial manifestation [J]. Journal of Peking University (Health Sciences), 2025, 57(6): 1061-1066. |
| [10] | Yajing GAO, Zhengfang LI, Mengsi MA, Lijun WU. Role of the SII and SIRI in risk prediction, disease activity assessment, and prog-nostic evaluation of Behçet disease uveitis [J]. Journal of Peking University (Health Sciences), 2025, 57(6): 1067-1073. |
| [11] | Xiaolin WANG, Shaoyi GUO, Dazhao CHEN, Xijie WEN, Yong HUA, Liang ZHANG, Qin ZHANG. A follow-up study on total hip arthroplasty in patients with systemic lupus erythematosus combined with osteonecrosis of femoral head [J]. Journal of Peking University (Health Sciences), 2025, 57(6): 1081-1088. |
| [12] | Xinfeng WU, Siliang MAN, Dazhao CHEN, Yong HUA, Xijie WEN, Yingyue DING, Liang ZHANG, Xiujuan HOU. A long-term review for radiographic parameters of hips in axial spondyloarthritis patients with end-stage hip involvement: A 24-year trend analysis from a single high-volume tertiary center [J]. Journal of Peking University (Health Sciences), 2025, 57(6): 1089-1095. |
| [13] | Jingwen LAN, Zhe CHEN, Yongjing CHENG, Like ZHAO. Clinical characteristics, efficacy and safety of antifibrotic agents in elderly patients with connective tissue disease-associated interstitial lung disease [J]. Journal of Peking University (Health Sciences), 2025, 57(6): 1101-1106. |
| [14] | Yanyan LI, Xiangjun LIU, Gongming LI, Chun LI, Yuan JIA. Clinic behavior and therapeutic status of patients with antiphospholipid syndrome [J]. Journal of Peking University (Health Sciences), 2025, 57(6): 1107-1112. |
| [15] | Chun WEI, Yue YANG, Xinju ZHAO, Xu LIU, Yuan JIA. Systemic lupus erythematosus complicated by autoimmune nodopathy: A case report [J]. Journal of Peking University (Health Sciences), 2025, 57(6): 1174-1179. |
|
||