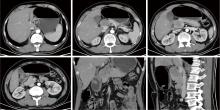
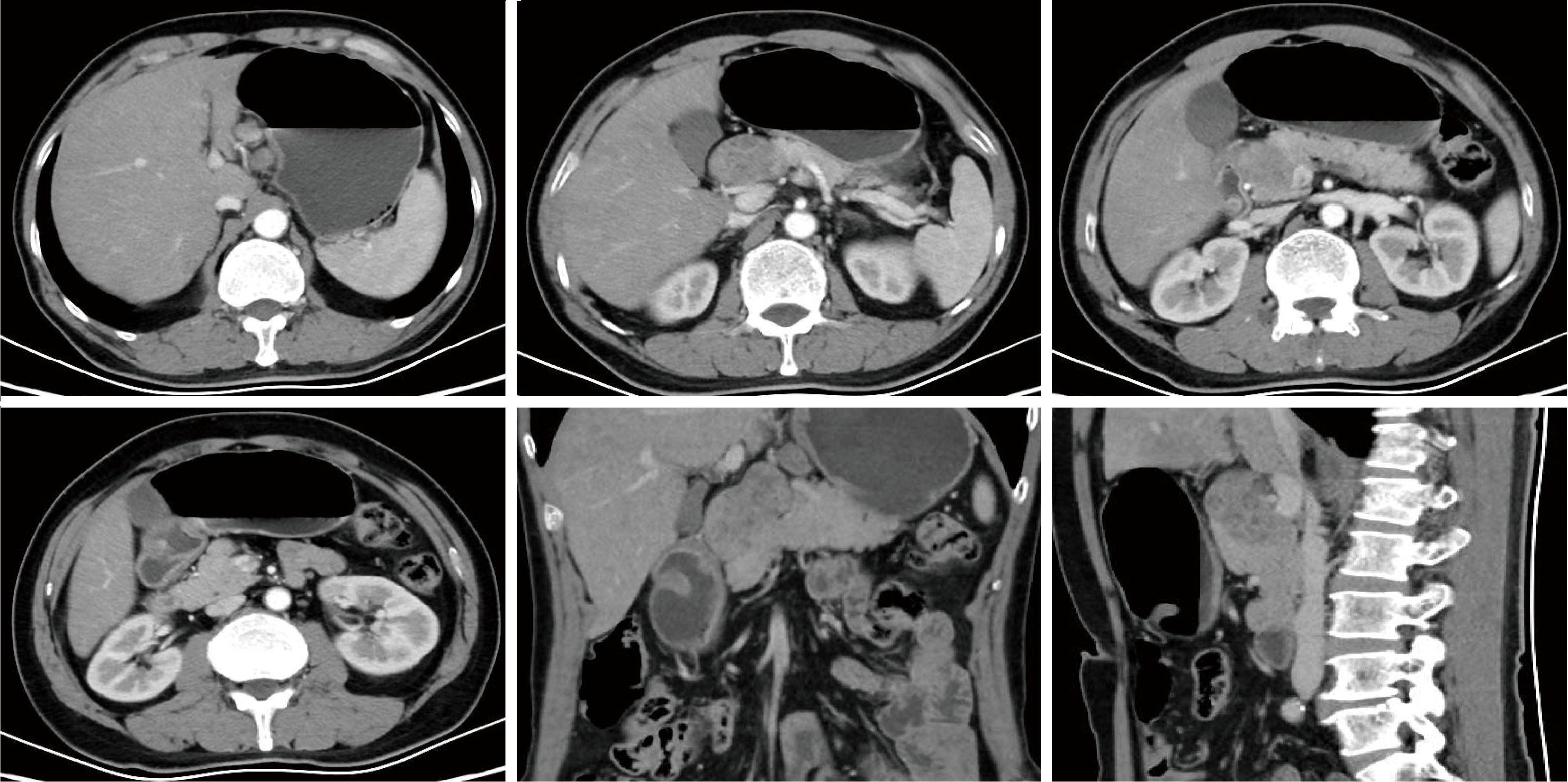
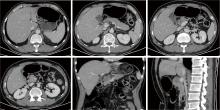
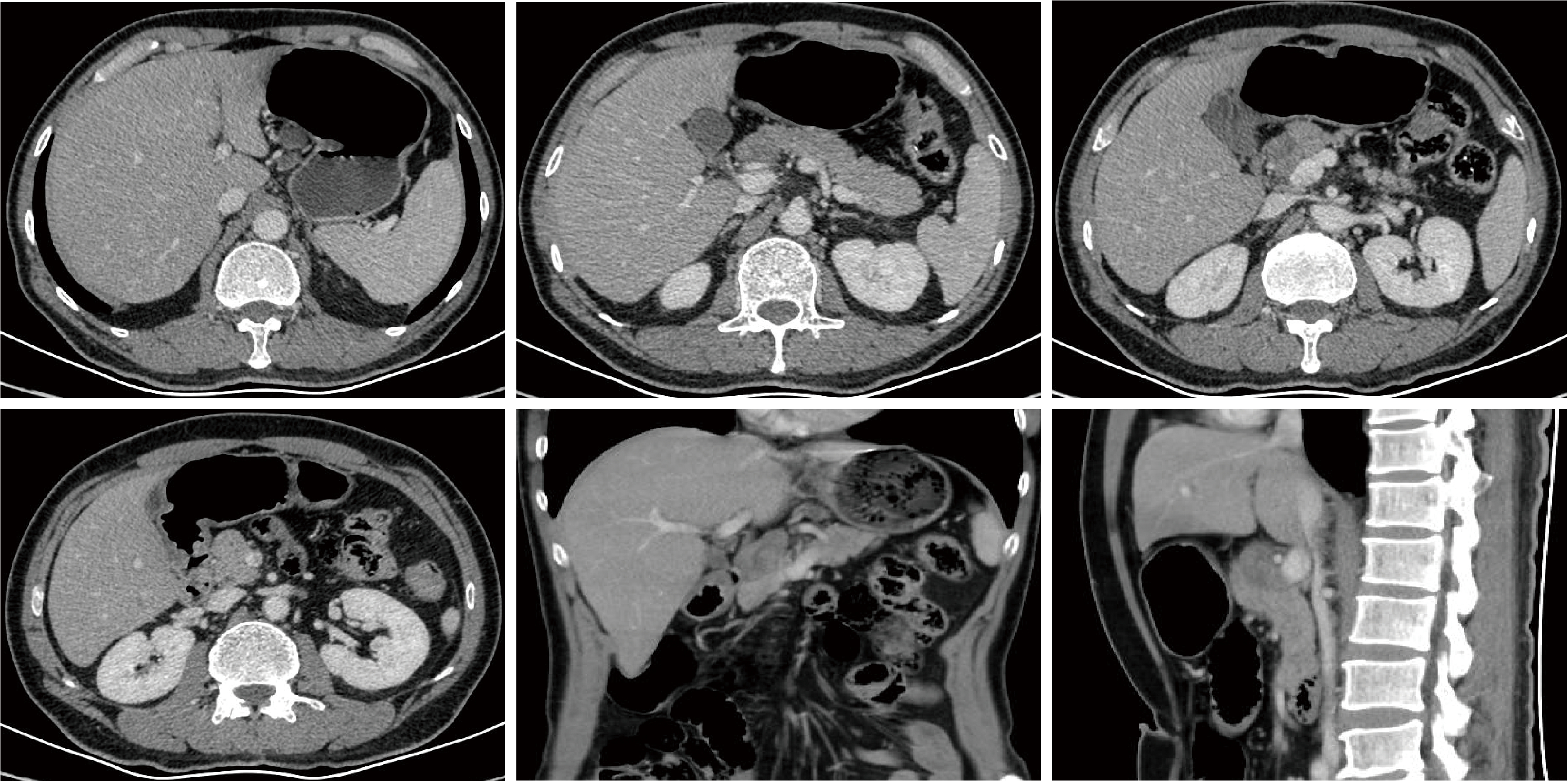
Journal of Peking University (Health Sciences) ›› 2026, Vol. 58 ›› Issue (2): 399-404. doi: 10.19723/j.issn.1671-167X.2026.02.027
Previous Articles Next Articles
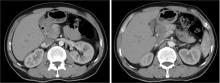
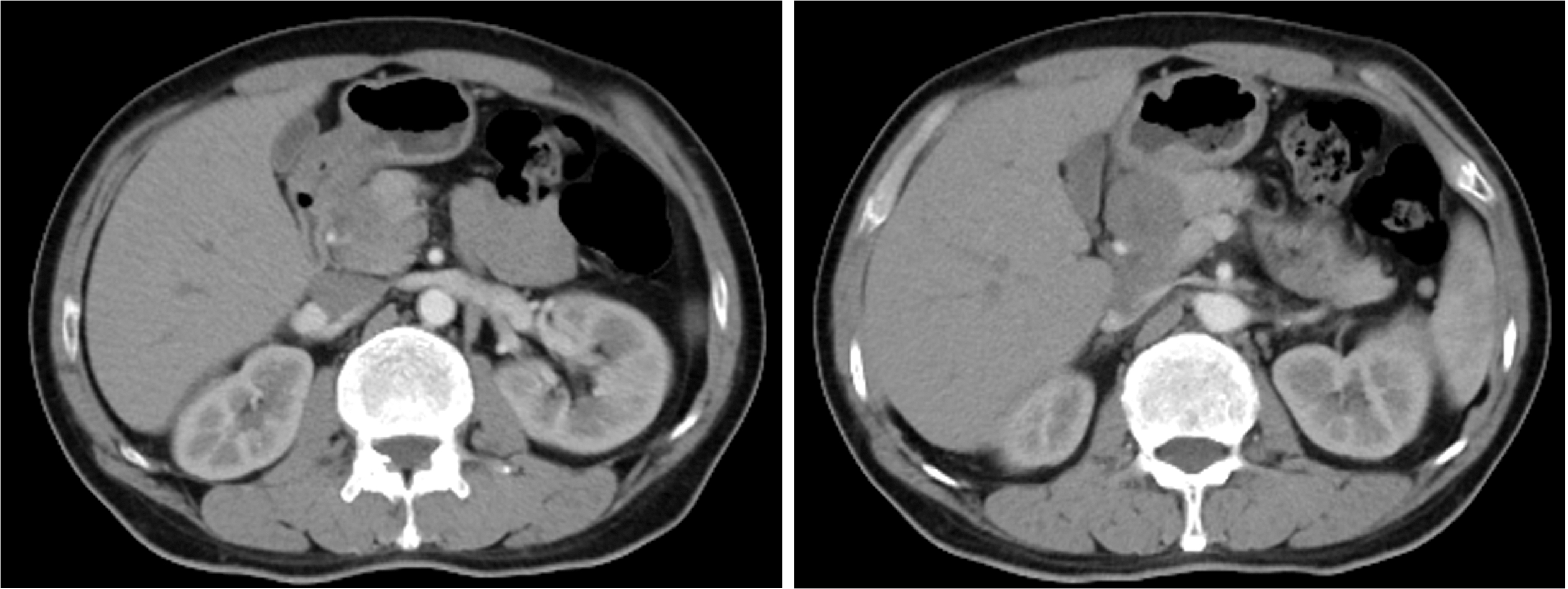
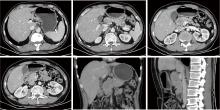
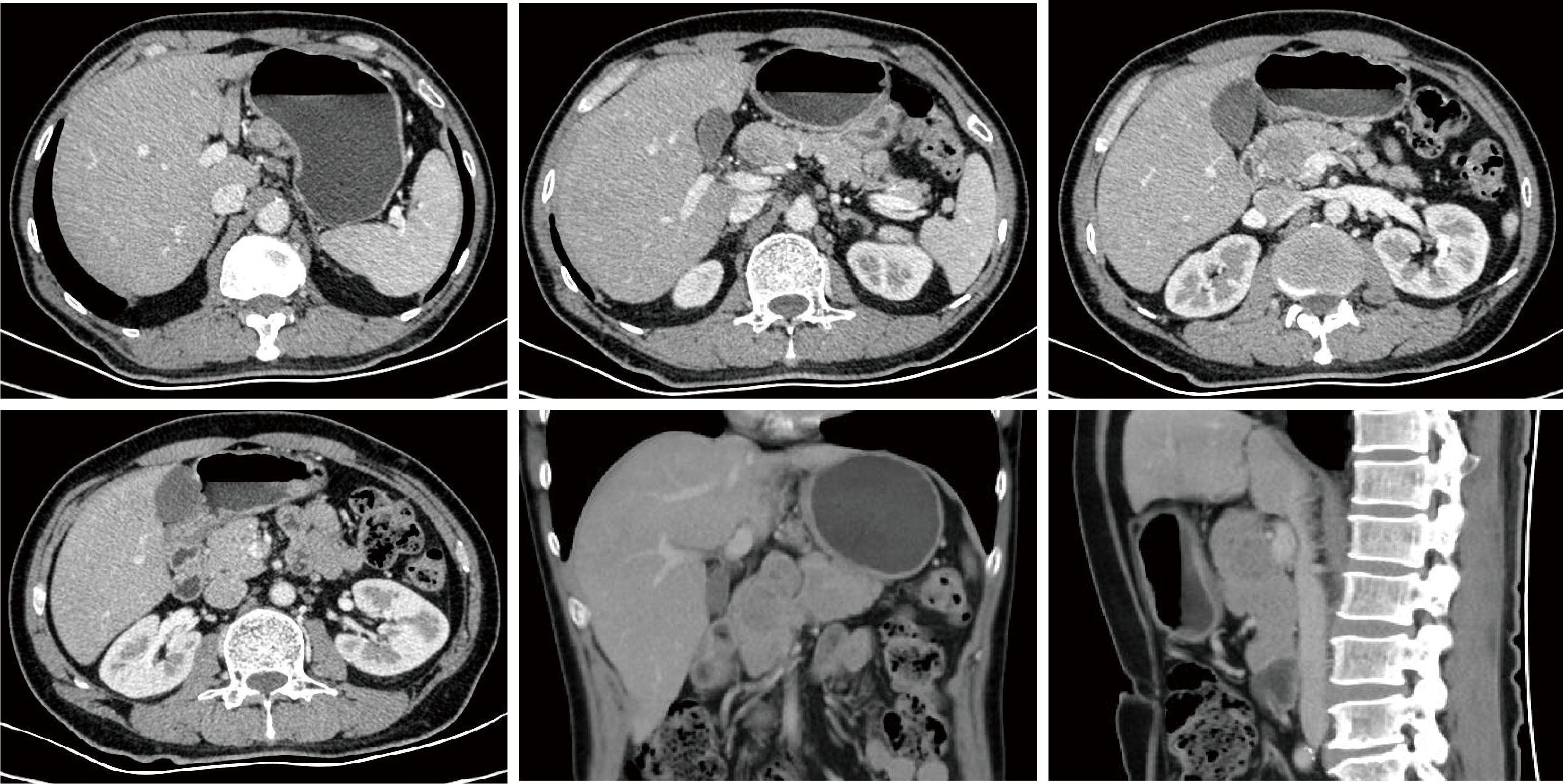
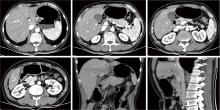
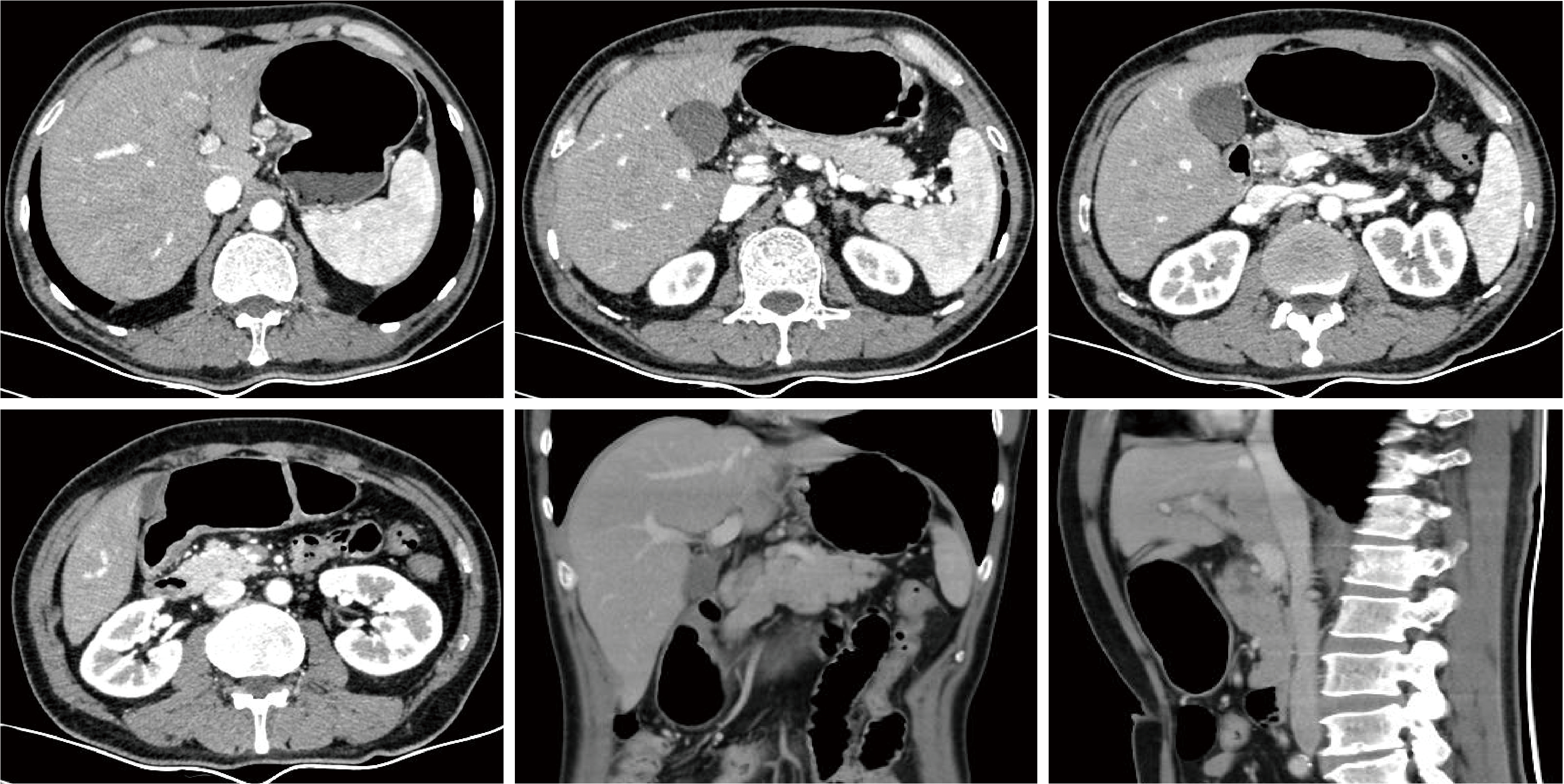
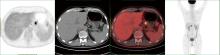
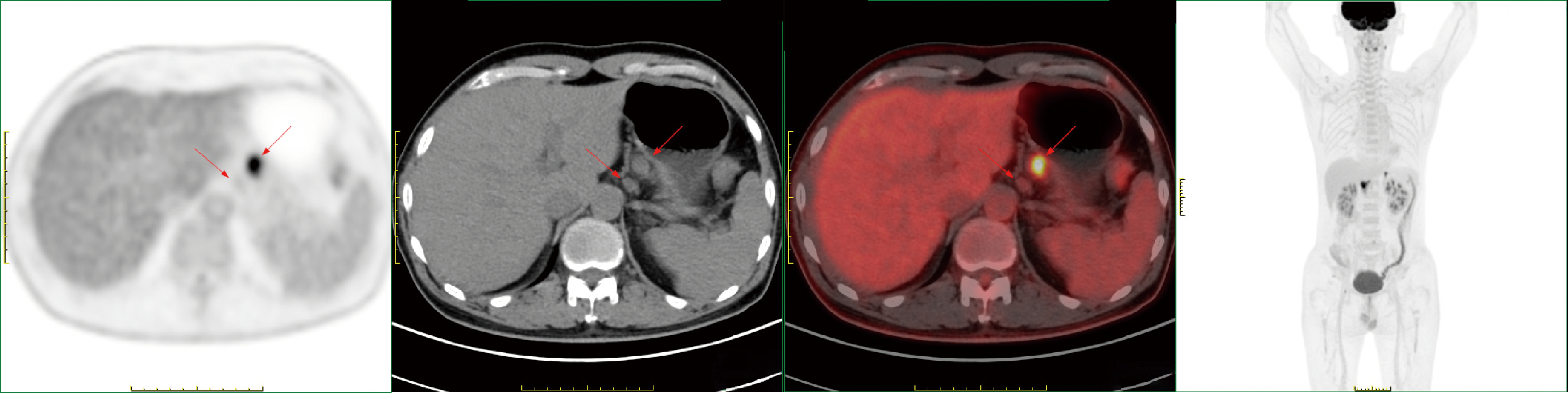
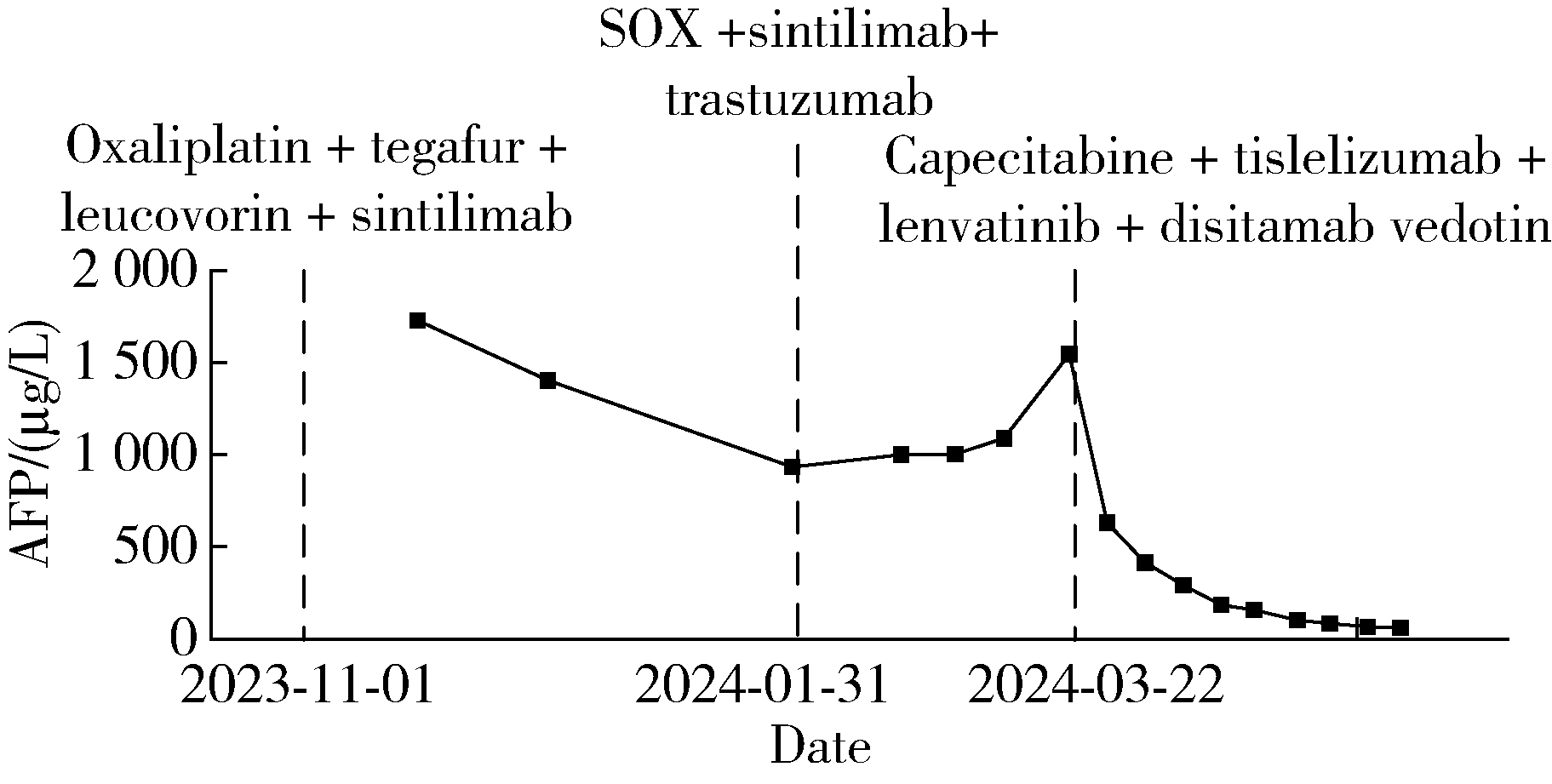
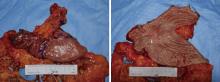
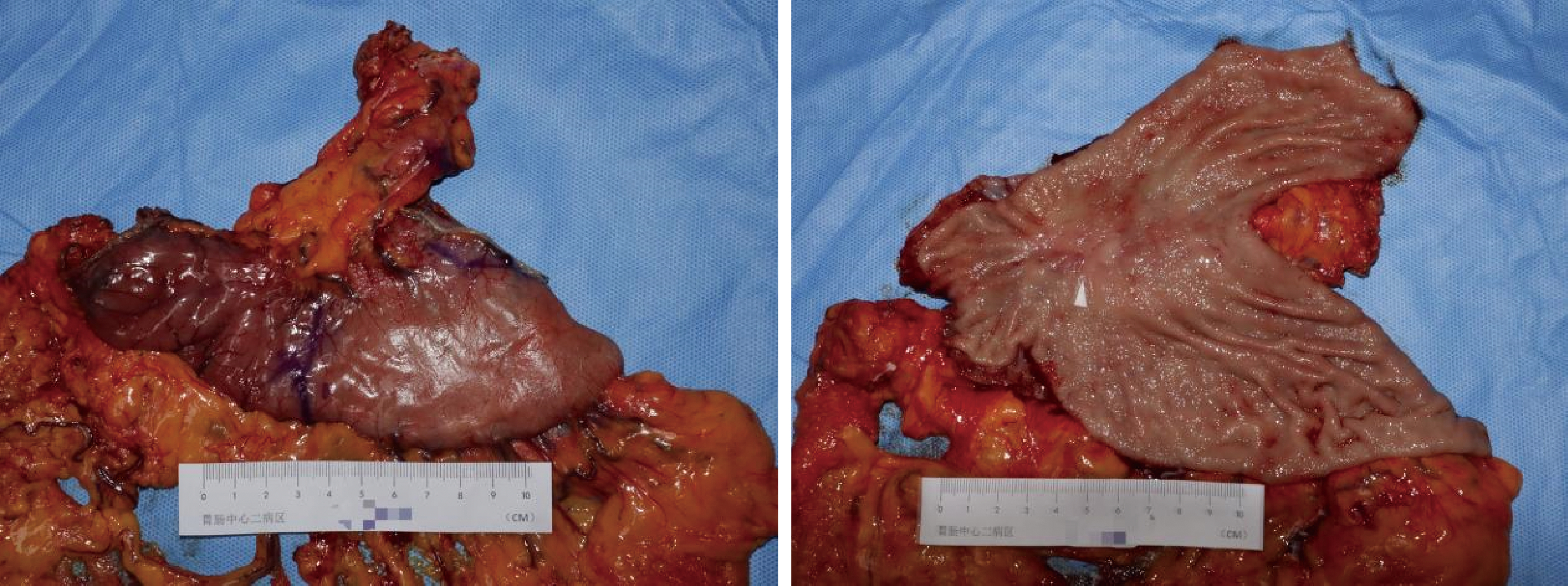
Conversion therapy for hepatoid adenocarcinoma of the stomach: A case report
Jialin LI1, Liqiao CHEN1, Jiatian TANG1, Yan WU2, Anqiang WANG1,*( )
)
- 1. Key laboratory of Carcinogenesis and Translational Research (Ministry of Education), Center of Gastrointestinal Cancer, Peking University Cancer Hospital & Institute, Beijing 100142, China
2. Key laboratory of Carcinogenesis and Translational Research (Ministry of Education), Department of Pathology, Peking University Cancer Hospital & Institute, Beijing 100142, China
CLC Number:
- R735.2
| 1 |
doi: 10.1002/cac2.12336 |
| 2 |
doi: 10.1038/s41388-021-01976-2 |
| 3 |
doi: 10.1158/0008-5472.CAN-07-5962 |
| 4 |
doi: 10.1007/s10549-015-3503-3 |
| 5 |
doi: 10.1002/cac2.12214 |
| 6 |
doi: 10.1007/s40265-019-01116-x |
| 7 |
doi: 10.1158/1078-0432.CCR-20-2571 |
| 8 |
doi: 10.1186/s12885-022-09405-7 |
| 9 |
doi: 10.1016/S1470-2045(20)30271-0 |
| [1] | Jiafu JI, Jingtao WEI, Ke JI, Zhaode BU. Bottlenecks and breakthroughs in gastric cancer diagnosis and treatment: Towards a new era of precision and intelligent integration [J]. Journal of Peking University (Health Sciences), 2026, 58(2): 231-238. |
| [2] | Jiale GAO, Zhongtao ZHANG. Current status and future perspectives of precision treatment for locally advanced rectal cancer [J]. Journal of Peking University (Health Sciences), 2026, 58(2): 247-250. |
| [3] | Hai WANG, Yizhou JIANG. Molecular mechanisms and clinical applications of anti-angiogenic therapy in precision treatment of breast cancer [J]. Journal of Peking University (Health Sciences), 2026, 58(2): 251-256. |
| [4] | Youdong LIU, Yajun LYU, Jie CHEN, Mingde ZANG, Hongda PAN, Xiaowen LIU, Jun LU, Fenglin LIU. Clinical efficacy and safety of totally laparoscopic subtotal gastrectomy with cardia-gastric fundus preservation in middle-upper gastric cancer [J]. Journal of Peking University (Health Sciences), 2026, 58(2): 301-306. |
| [5] | Bin LI, Han LIANG. Robotic gastrectomy: Research progress and practical challenges [J]. Journal of Peking University (Health Sciences), 2026, 58(2): 416-422. |
| [6] | Xiaolin WANG, Luyao LI, Wen ZHANG, Hongyan WANG. Clinicopathological analysis of mesonephric-like adenocarcinoma in the corpusuteri: A report of 3 cases [J]. Journal of Peking University (Health Sciences), 2025, 57(6): 1208-1212. |
| [7] | Weihua HOU, Shujie SONG, Zhongyue SHI, Lu LIU, Mulan JIN. Neuroendocrine carcinoma with significantly vacuolar nucleus at the esophagogastric junction: A case report [J]. Journal of Peking University (Health Sciences), 2025, 57(5): 1005-1009. |
| [8] | Shang XIE, Luming WANG, Xinyuan ZHANG, Qiushi FENG, Yangyang XIA, Ziwei DAI, Xiaofeng SHAN, Zhigang CAI. Construction and application of oral squamous cell carcinoma organoid bank [J]. Journal of Peking University (Health Sciences), 2025, 57(5): 847-851. |
| [9] | Xiaoyong YANG, Fan ZHANG, Lulin MA, Cheng LIU. Clinical characteristics and influencing factors of extraglandular invasion of prostatic ductal adenocarcinoma [J]. Journal of Peking University (Health Sciences), 2025, 57(5): 956-960. |
| [10] | Dan LUO, Haijian HUANG, Xin CHEN, Xiaoyan CHEN. Primary uterine hepatoid adenocarcinoma: Clinicopathological analysis of 2 cases and literature review [J]. Journal of Peking University (Health Sciences), 2024, 56(6): 1126-1131. |
| [11] | Shang XIE,Zhi-gang CAI,Xiao-feng SHAN. Application value of whole exon sequencing and immune related indicators in the precision treatment of oral squamous cell carcinoma [J]. Journal of Peking University (Health Sciences), 2023, 55(4): 697-701. |
| [12] | Qi SHEN,Yi-xiao LIU,Qun HE. Mucinous tubular and spindle cell carcinoma of kidney: Clinicopathology and prognosis [J]. Journal of Peking University (Health Sciences), 2023, 55(2): 276-282. |
| [13] | Wei-hua HOU,Shu-jie SONG,Zhong-yue SHI,Mu-lan JIN. Clinicopathological features of Helicobacter pylori-negative early gastric cancer [J]. Journal of Peking University (Health Sciences), 2023, 55(2): 292-298. |
| [14] | Yan XIONG,Bo ZHANG,Li-gong NIE,Shi-kai WU,Hu ZHAO,Dong LI,Ji-ting DI. Thoracic SMARCA4-deficient undifferentiated tumor-pathological diagnosis and combined immune checkpoint inhibitor treatment [J]. Journal of Peking University (Health Sciences), 2023, 55(2): 351-356. |
| [15] | Bo-han NING,Qing-xia ZHANG,Hui YANG,Ying DONG. Endometrioid adenocarcinoma with proliferated stromal cells, hyalinization and cord-like formations: A case report [J]. Journal of Peking University (Health Sciences), 2023, 55(2): 366-369. |
|
||