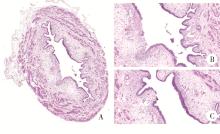
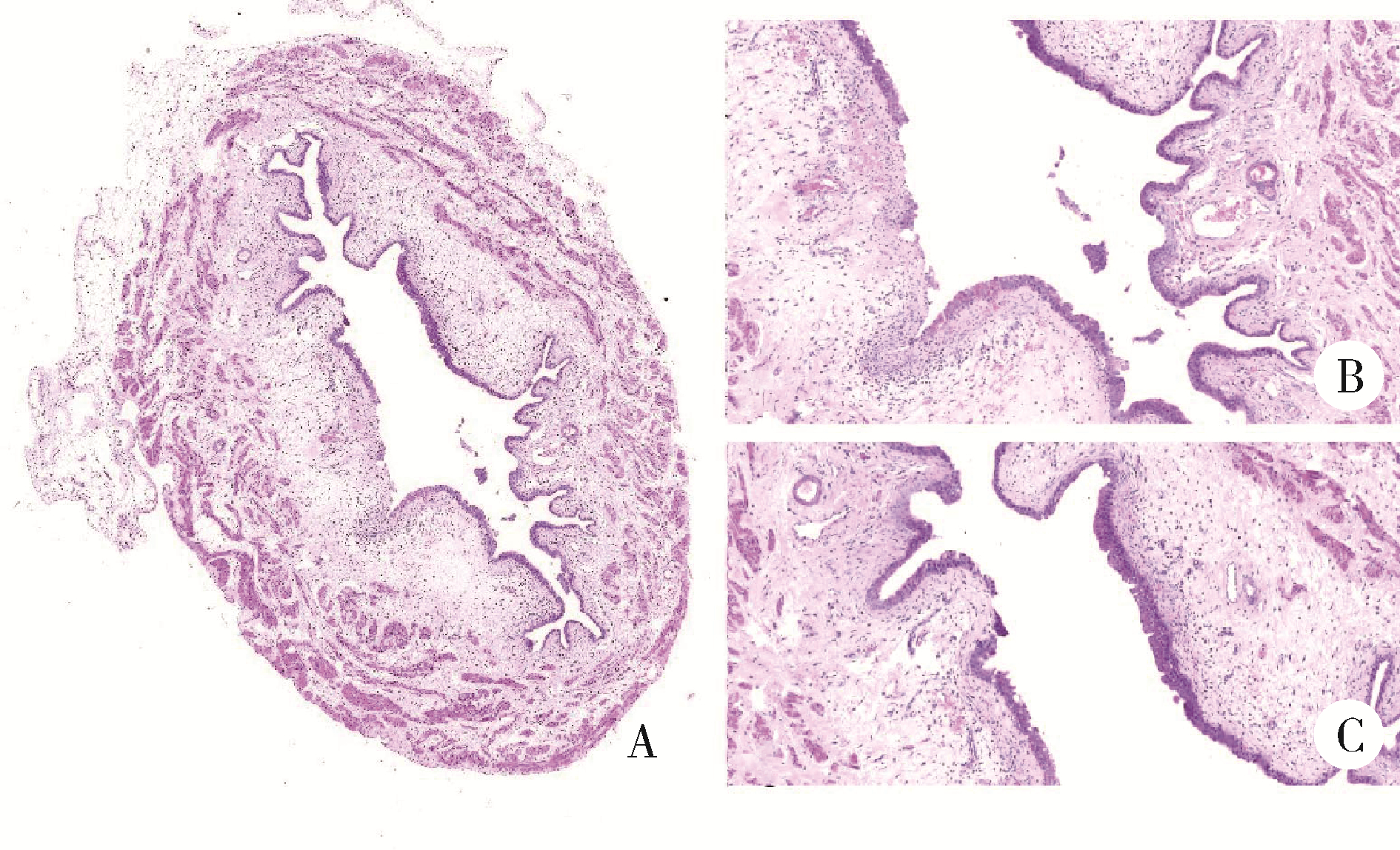
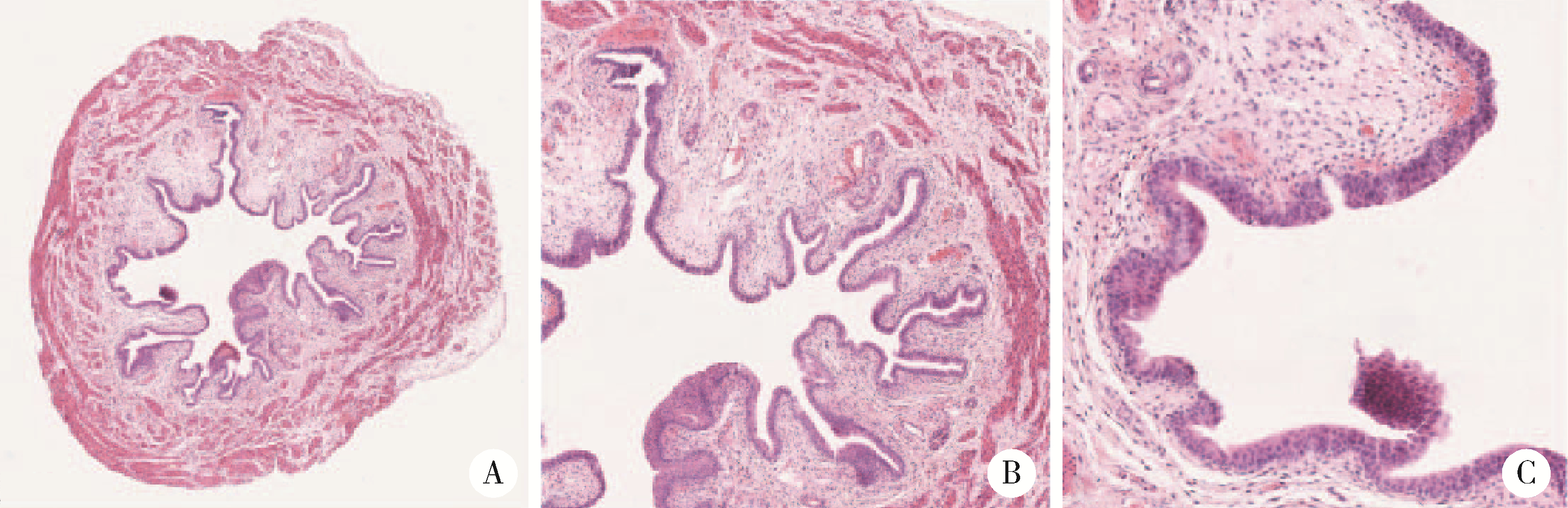
Journal of Peking University (Health Sciences) ›› 2022, Vol. 54 ›› Issue (4): 735-740. doi: 10.19723/j.issn.1671-167X.2022.04.024
Previous Articles Next Articles
Urodynamic and histological evaluation of cyclophosphamide-induced bladder pain syndrome in SD rats
Lin ZHU,Wei-yu ZHANG,Ke-xin XU*( )
)
- Department of Urology, Peking University People's Hospital, Beijing 100044, China
CLC Number:
- R694.5
| 1 | Hakimi Z , Houbiers J , Pedersini R , et al. The burden of bladder pain in five European countries: a cross-sectional study[J]. Urology, 2017, 99 (1): 84- 91. |
| 2 |
Hanno P , Lin A , Nordling J , et al. Bladder pain syndrome committee of the International Consultation on Incontinence[J]. Neurourol Urodyn, 2010, 29 (1): 191- 198.
doi: 10.1002/nau.20847 |
| 3 |
Homma Y , Akiyama Y , Tomoe H , et al. Clinical guidelines for interstitial cystitis/bladder pain syndrome[J]. Int J Urol, 2020, 27 (7): 578- 589.
doi: 10.1111/iju.14234 |
| 4 | Akiyama Y , Hanno P . Phenotyping of interstitial cystitis/bladder pain syndrome[J]. Int J Urol, 2019, 26 (Suppl 1): 17- 19. |
| 5 | Chai TC , Russo A , Yu S , et al. Mucosal signaling in the bladder[J]. Auton Neurosci, 2016, 200 (10): 49- 56. |
| 6 |
Karamali M , Shafabakhsh R , Ghanbari Z , et al. Molecular pathogenesis of interstitial cystitis/bladder pain syndrome based on gene expression[J]. J Cell Physiol, 2019, 234 (8): 12301- 12308.
doi: 10.1002/jcp.28009 |
| 7 | Lee MH , Wu HC , Tseng CM , et al. Health education and symptom flare management using a video-based health system for caring women with BPS/IC[J]. Urology, 2018, 119 (9): 62- 69. |
| 8 | Crescenze IM , Tucky B , Li J , Moore C , et al. Efficacy, side effects, and monitoring of oral cyclosporine in interstitial cystitis-bladder pain syndrome[J]. Urology, 2017, 107 (9): 49- 54. |
| 9 |
杨进益, 魏伟, 叶林, 等. 膀胱水扩张后透明质酸钠灌注治疗间质性膀胱炎疗效分析[J]. 中华泌尿外科杂志, 2012, 33 (3): 219- 222.
doi: 10.3760/cma.j.issn.1000-6702.2012.03.019 |
| 10 |
Ryu CM , Yu HY , Lee HY , et al. Longitudinal intravital imaging of transplanted mesenchymal stem cells elucidates their functional integration and therapeutic potency in an animal model of interstitial cystitis/bladder pain syndrome[J]. Theranostics, 2018, 8 (20): 5610- 5624.
doi: 10.7150/thno.27559 |
| 11 |
Lv YS , Yao YS , Rong L , et al. Intravesical hyaluronidase causes chronic cystitis in a rat model: a potential model of bladder pain syndrome/interstitial cystitis[J]. Int J Urol, 2014, 21 (6): 601- 607.
doi: 10.1111/iju.12358 |
| 12 |
Mills KA , Chess-Williams R , McDermott C . Novel insights into the mechanism of cyclophosphamide-induced bladder toxicity: chloroacetaldehyde's contribution to urothelial dysfunction in vitro[J]. Arch Toxicol, 2019, 93 (11): 3291- 3303.
doi: 10.1007/s00204-019-02589-1 |
| 13 |
de Oliveira MG , Mónica FZ , Calmasini FB , et al. Deletion or pharmacological blockade of TLR4 confers protection against cyclophosphamide-induced mouse cystitis[J]. Am J Physiol Renal Physiol, 2018, 315 (3): 460- 468.
doi: 10.1152/ajprenal.00100.2018 |
| 14 | Augé C , Gamé X , Vergnolle N , et al. Characterization and validation of a chronic model of cyclophosphamide-induced interstitial cystitis/bladder pain syndrome in rats[J]. Front Pharmacol, 2020, 11 (8): 1305. |
| 15 |
Yang W , Yaggie RE , Jiang MC , et al. Acyloxyacyl hydrolase modulates pelvic pain severity[J]. Am J Physiol Regul Integr Comp Physiol, 2018, 314 (3): 353- 365.
doi: 10.1152/ajpregu.00239.2017 |
| 16 | Lee UJ , Ackerman AL , Wu A , Zhang R , et al. Chronic psychological stress in high-anxiety rats induces sustained bladder hyperalgesia[J]. Physiol Behav, 2015, 139 (2): 541- 548. |
| 17 |
Akiyama Y , Luo Y , Hanno PM , et al. Interstitial cystitis/bladder pain syndrome: the evolving landscape, animal models and future perspectives[J]. Int J Urol, 2020, 27 (6): 491- 503.
doi: 10.1111/iju.14229 |
| 18 |
Birder L , Andersson KE . Animal modelling of interstitial cystitis/bladder pain syndrome[J]. Int Neurourol J, 2018, 22 (Suppl 1): 3- 9.
doi: 10.5213/inj.1835062.531 |
| [1] | Hanwei KE, Qi WANG, Kexin XU. Optimization study of an animal model for interstitial cystitis/bladder pain syndrome based on the dose effect of cyclophosphamide [J]. Journal of Peking University (Health Sciences), 2024, 56(5): 908-912. |
| [2] | Peng XIN,Hao ZHANG,Zhen-ming JIANG. Treatment of intravesical instillation with fulguration-hydrodistention on female interstitial cystitis [J]. Journal of Peking University (Health Sciences), 2023, 55(5): 865-870. |
| [3] | Ling-wei MENG,Xue LI,Sheng-han GAO,Yue LI,Rui-tao CAO,Yi ZHANG,Shao-xia PAN. Comparison of three methods for establishing rat peri-implantitis model [J]. Journal of Peking University (Health Sciences), 2023, 55(1): 22-29. |
| [4] | Miao SHAO,Hui-fang GUO,Ling-yan LEI,Qing ZHAO,Yan-jie DING,Jin LIN,Rui WU,Feng YU,Yu-cui LI,Hua-li MIAO,Li-yun ZHANG,Yan DU,Rui-ying JIAO,Li-xia PANG,Li LONG,Zhan-guo LI,Ru LI. A multicenter study on the tolerance of intravenous low-dose cyclophosphamide in systemic lupus erythematosus [J]. Journal of Peking University (Health Sciences), 2022, 54(6): 1112-1116. |
| [5] | WANG Gui-hong,ZUO Ting,LI Ran,ZUO Zheng-cai. Effect of rebamipide on the acute gouty arthritis in rats induced by monosodium urate crystals [J]. Journal of Peking University (Health Sciences), 2021, 53(4): 716-720. |
| [6] | WANG Jia-wen,LIU Jing-chao,MENG Ling-feng,ZHANG Wei,LIU Xiao-dong,ZHANG Yao-guang. Quality of life and related factors in patients with interstitial cystitis/bladder pain syndrome [J]. Journal of Peking University (Health Sciences), 2021, 53(4): 653-658. |
| [7] | Tao WANG,Ke-xin XU,Wei-yu ZHANG,Hao HU,Xiao-wei ZHANG,Huan-rui WANG,Xian-hui LIU,Jing-wen CHEN,Xiao-peng ZHANG. Urodynamic classification of male patients with symptoms of overactive bladder and the outcome classification [J]. Journal of Peking University(Health Sciences), 2019, 51(6): 1048-1051. |
| [8] | Wei-yu ZHANG,Qiu-xiang XIA,Hao HU,Jing-wen CHEN,Yi-ran SUN,Ke-xin XU,Xiao-peng ZHANG. Analysis of urodynamic study of female outpatients with lower urinary tract symptoms and follow-up of the patients with detrusor underactive [J]. Journal of Peking University(Health Sciences), 2019, 51(5): 856-862. |
| [9] | ZHANG Wei-yu, ZHANG Xiao-peng, CHEN Jing-wen, SUN Yi-ran, WANG Jia, HU Hao, XU Ke-xin. Effect of age on urodynamic parameters of women with urinary incontinence [J]. Journal of Peking University(Health Sciences), 2016, 48(5): 825-829. |
| [10] | ZHANG Wei-yu, HU Hao, WANG Qi, CHEN Jing-wen, XU Ke-xin. Significance of preoperative urodynamics for clinical diagnosis of female patients with stress urinary incontinence [J]. Journal of Peking University(Health Sciences), 2016, 48(4): 655-658. |
| Viewed | ||||||||||||||||||||||||||||||||||||||||||||||||||
|
Full text 199
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||
|
Abstract 573
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||
Cited |
|
|||||||||||||||||||||||||||||||||||||||||||||||||
| Shared | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Discussed | ||||||||||||||||||||||||||||||||||||||||||||||||||
|
||