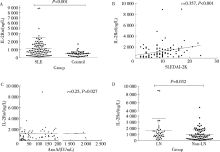
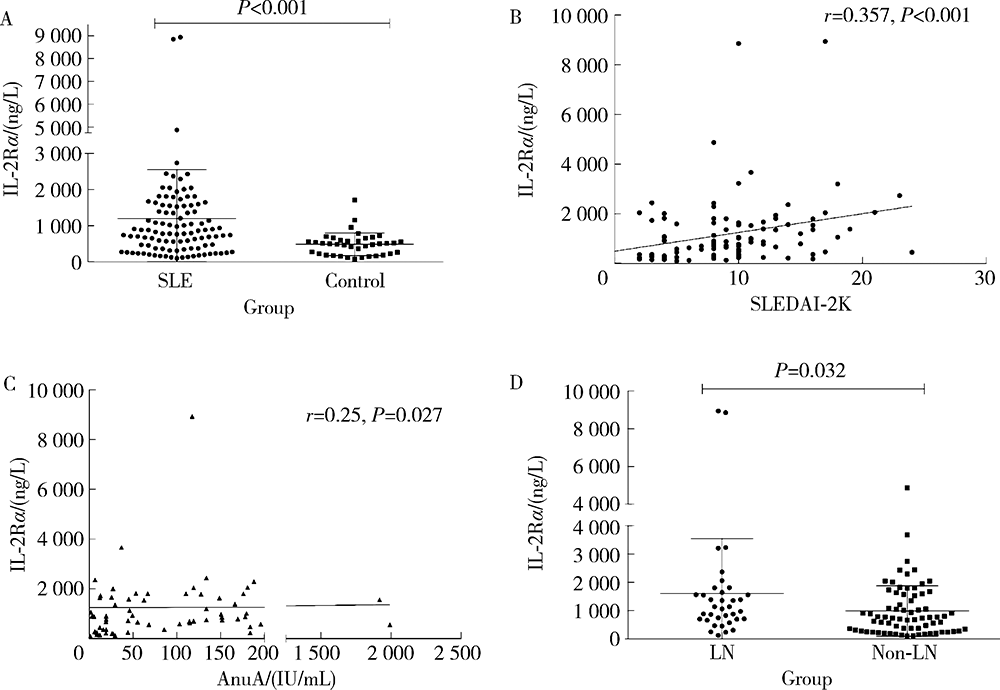
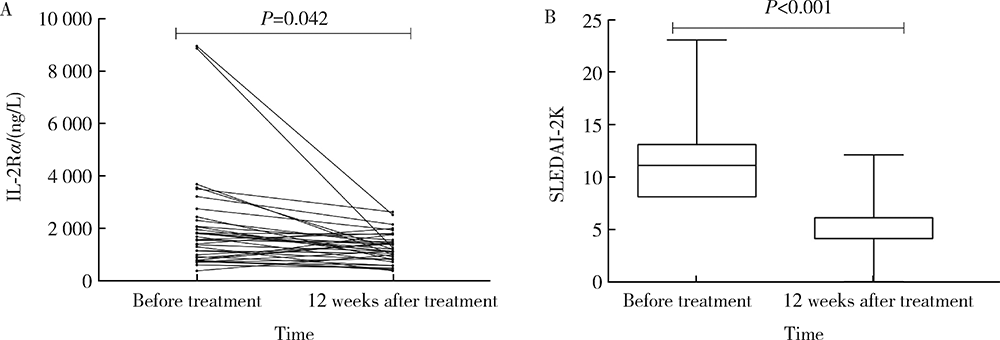
Journal of Peking University (Health Sciences) ›› 2021, Vol. 53 ›› Issue (6): 1083-1087. doi: 10.19723/j.issn.1671-167X.2021.06.013
Previous Articles Next Articles
Serum interleukin-2 receptor α as a clinical biomarker in patients with systemic lupus erythematosus
TIAN Jia-yi,ZHANG Xia( ),CHENG Gong,LIU Qing-hong,WANG Shi-yang,HE Jing(
),CHENG Gong,LIU Qing-hong,WANG Shi-yang,HE Jing( )
)
- Department of Rheumatology and Immunology, Peking University People’s Hospital, Beijing 100044, China
CLC Number:
- R593.24
| [1] |
Tsokos GC. Autoimmunity and organ damage in systemic lupus erythematosus[J]. Nat Immunol, 2020, 21(6):605-614.
doi: 10.1038/s41590-020-0677-6 |
| [2] |
Ross SH, Cantrell DA. Signaling and function of interleukin-2 in T lymphocytes[J]. Annu Rev Immunol, 2018, 36:411-433.
doi: 10.1146/immunol.2018.36.issue-1 |
| [3] | He J, Zhang R, Shao M, et al. Efficacy and safety of low-dose IL-2 in the treatment of systemic lupus erythematosus: A randomised, double-blind, placebo-controlled trial[J]. Ann Rheum Dis, 2020, 79(1):141-149. |
| [4] |
He J, Zhang X, Wei Y, et al. Low-dose interleukin-2 treatment selectively modulates CD4(+) T cell subsets in patients with systemic lupus erythematosus[J]. Nat Med, 2016, 22(9):991-993.
doi: 10.1038/nm.4148 |
| [5] |
Rubin L A, Galli F, Greene WC, et al. The molecular basis for the generation of the human soluble interleukin 2 receptor[J]. Cytokine, 1990, 2(5):330-336.
pmid: 2103332 |
| [6] | Dik WA, Heron M. Clinical significance of soluble interleukin-2 receptor measurement in immune-mediated diseases[J]. Neth J Med, 2020, 78(5):220-231. |
| [7] |
Luo H, Wang C, Feng M, et al. Microgravity inhibits resting T cell immunity in an exposure time-dependent manner[J]. Int J Med Sci, 2014, 11(1):87-96.
doi: 10.7150/ijms.7651 |
| [8] |
El-Shafey EM, El-Nagar GF, El-Bendary AS, et al. Serum soluble interleukin-2 receptor alpha in systemic lupus erythematosus[J]. Iran J Kidney Dis, 2008, 2(2):80-85.
pmid: 19377213 |
| [9] | Hochberg MC. Updating the American College of Rheumatology revised criteria for the classification of systemic lupus erythematosus[J]. Arthritis Rheum, 1997, 40(9):1725. |
| [10] |
Gladman DD, Ibañez D, Urowitz MB. Systemic lupus erythematosus disease activity index 2000[J]. J Rheumatol, 2002, 29(2):288-291.
pmid: 11838846 |
| [11] |
Romero-Diaz J, Isenberg D, Ramsey-Goldman R. Measures of adult systemic lupus erythematosus: Updated version of British Isles Lupus Assessment Group (BILAG 2004), European Consensus Lupus Activity Measurements (ECLAM), Systemic Lupus Activity Measure, Revised (SLAM-R), Systemic Lupus Activity Questionnaire for Population Studies (SLAQ), Systemic Lupus Erythematosus Disease Activity Index 2000 (SLEDAI-2K), and Systemic Lupus International Collaborating Clinics/American College of Rheumatology Damage Index (SDI)[J]. Arthritis Care Res (Hoboken), 2011, 63(Suppl 11):S37-S46.
doi: 10.1002/acr.v63.11s |
| [12] |
Russell SE, Moore AC, Fallon PG, et al. Soluble IL-2Rα (sCD25) exacerbates autoimmunity and enhances the development of Th17 responses in mice[J]. PLoS One, 2012, 7(10):e47748.
doi: 10.1371/journal.pone.0047748 |
| [13] | Spolski R, Li P, Leonard WJ. Biology and regulation of IL-2: From molecular mechanisms to human therapy[J]. Nat Rev Immunol, 2018, 18(10):648-659. |
| [14] | Mizui M, Tsokos GC. Targeting regulatory T cells to treat patients with systemic lupus erythematosus[J]. Front Immunol, 2018(9):786. |
| [15] |
Laut J, Senitzer D, Petrucci R, et al. Soluble interleukin-2 receptor levels in lupus nephritis[J]. Clin Nephrol, 1992, 38(4):179-184.
pmid: 1424303 |
| [1] | Xiaofei TANG, Qiuling DING, Yang ZHANG, Yumei WANG, Meiyi TIAN, Zhuo SUN, Rongrong WANG, Yan CHEN, Jian LIU. Refractory systemic lupus erythematosus-associated thrombocytopenia treated with avatrombopag: A case report [J]. Journal of Peking University (Health Sciences), 2026, 58(2): 405-409. |
| [2] | Yajing GAO, Zhengfang LI, Mengsi MA, Lijun WU. Role of the SII and SIRI in risk prediction, disease activity assessment, and prog-nostic evaluation of Behçet disease uveitis [J]. Journal of Peking University (Health Sciences), 2025, 57(6): 1067-1073. |
| [3] | Xinyi LI, Jinxia ZHAO, Rong MU. Impact of aspirin use on pregnancy outcomes in patients with systemic lupus erythematosus [J]. Journal of Peking University (Health Sciences), 2025, 57(6): 1074-1080. |
| [4] | Xiaolin WANG, Shaoyi GUO, Dazhao CHEN, Xijie WEN, Yong HUA, Liang ZHANG, Qin ZHANG. A follow-up study on total hip arthroplasty in patients with systemic lupus erythematosus combined with osteonecrosis of femoral head [J]. Journal of Peking University (Health Sciences), 2025, 57(6): 1081-1088. |
| [5] | Chun WEI, Yue YANG, Xinju ZHAO, Xu LIU, Yuan JIA. Systemic lupus erythematosus complicated by autoimmune nodopathy: A case report [J]. Journal of Peking University (Health Sciences), 2025, 57(6): 1174-1179. |
| [6] | Jingyuan LIANG, Xia ZHANG, Haihong YAO. POEMS syndrome misdiagnosed as systemic sclerosis: A case report [J]. Journal of Peking University (Health Sciences), 2025, 57(6): 1184-1187. |
| [7] | Kai ZHAO, Fu'ai LU, Yongfu WANG. Central nervous system infection mimicking neuropsychiatric systemic lupus erythematosus: A case report [J]. Journal of Peking University (Health Sciences), 2025, 57(6): 1188-1192. |
| [8] | Pei CAO, Qingxian LUAN. Periodontitis and systemic diseases: Thinking and explorations [J]. Journal of Peking University (Health Sciences), 2025, 57(5): 852-858. |
| [9] | Lixiu ZHU, Renli CHEN, Sujuan ZHOU, Ye LIN, Yirong TANG, Zhen YE. Effect of aquaporin 5 on TLR4/MyD88/NF-κB signaling pathway in Sjögren syndrome rats [J]. Journal of Peking University (Health Sciences), 2025, 57(5): 875-883. |
| [10] | Xiandun YUAN, Zhaohua LI, Dan XU, Ting LI, Dan FANG, Rong MU. Pathogenesis and mechanism of serine protease 23 in skin fibrosis of systemic sclerosis [J]. Journal of Peking University (Health Sciences), 2025, 57(5): 903-910. |
| [11] | Wenqiong WANG, Yuke HOU, Chun LI, Xuewu ZHANG. Predictors of adverse pregnancy outcomes in patients with systemic lupus erythematosus [J]. Journal of Peking University (Health Sciences), 2025, 57(3): 599-603. |
| [12] | Mengyue HUO, Hua MEI, Yuheng ZHANG, Yanbo ZHANG, Chunli LIU. Expression and regulatory mechanism of miR-34a in neonatal rat model of bron-chopulmonary dysplasia induced by hyperoxia [J]. Journal of Peking University (Health Sciences), 2025, 57(2): 237-244. |
| [13] | Hongyan WANG, Xinming LI, Kechi FANG, Huaqun ZHU, Rulin JIA, Jing WANG. Analysis of characteristics related to the disease activity of systemic lupus erythematosus and construction of an evaluation model [J]. Journal of Peking University (Health Sciences), 2024, 56(6): 1017-1022. |
| [14] | Dandan CHEN, Yun LI, Qingyi LU, Xiaohong XIANG, Feng SUN, Yingni LI, Jing ZHAO, Hongyan WANG, Chun LI. Ovarian function in patients of childbearing age with systemic lupus erythematosus [J]. Journal of Peking University (Health Sciences), 2024, 56(6): 1023-1028. |
| [15] | Li WANG, Chao GAO, Huanhuan REN, Yanping SHEN, Xiaowei HUANG, Hong YAO, Dandan HAN. Current status and influential factors of self-management ability in patients with systemic lupus erythematosus [J]. Journal of Peking University (Health Sciences), 2024, 56(6): 1029-1035. |
|
||