Journal of Peking University (Health Sciences) ›› 2020, Vol. 52 ›› Issue (3): 570-577. doi: 10.19723/j.issn.1671-167X.2020.03.026
Previous Articles Next Articles
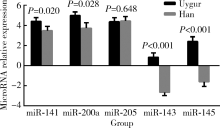
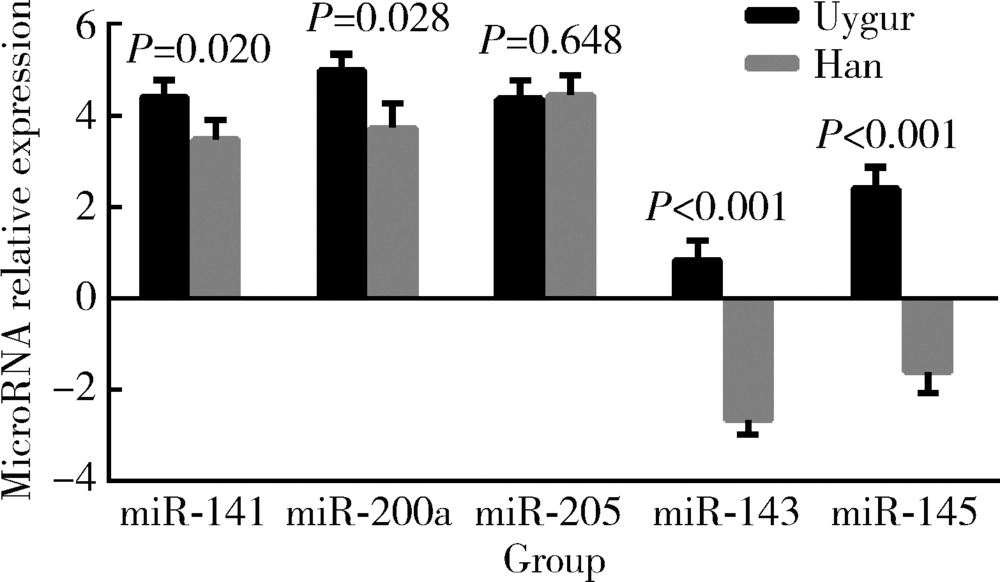
Characteristic and clinical significance of microRNA expression between 144 Uygur and Han women with endometrial carcinoma
Xiao WANG1,Dan HE1,Wen-ting LI2,SIYITI Adila·2,Rui HAN1,Ying DONG1,△( )
)
- 1. Department of Pathology, Peking University First Hospital, Beijing 100034, China
2. Department of Pathology, Affiliated Cancer Hospital of Xinjiang Medical University, Urumqi 830011, China
CLC Number:
- R737.33
| [1] | 廖秦平, 杨曦. 子宫内膜癌筛查及早期诊断的现状及展望. 实用妇产科杂志, 2015,31(7):481-484. |
| [2] | Cancer Genome Atlas Research, Network. Integrated genomic characterization of endometrial carcinoma[J]. Nature, 2013,497(7447):67-73. |
| [3] | Schimp VL, Ali-Fehmi R, Solomon LA, et al. The racial disparity in outcomes in endometrial cancer: could this be explained on a molecular level?[J]. Gynecol Oncol, 2006,102(3):440-446. |
| [4] | Srivastava SK, Ahmad A, Zubair H, et al. MicroRNAs in gynecological cancers: Small molecules with big implications[J]. Cancer Lett, 2017,407:123-138. |
| [5] | Robert JK, Maria LC, Herrington CS, et al. WHO classification of tumours of female reproductive organs[M]. Lyon, France: World Health Organization, 2014: 121-154. |
| [6] | 董颖, 周梅, 巴晓军, 等. 子宫内膜癌中DNA甲基转移酶3B的表达特点与临床意义[J]. 北京大学学报(医学版), 2016,48(5):788-794. |
| [7] | Chen T, Ding L, Shan G, et al. Prevalence and racial differences in pterygium: a cross-sectional study in Han and Uygur adults in Xinjiang, China[J]. Invest Ophthalmol Vis Sci, 2015,56(2):1109-1117. |
| [8] | Lee JW, Park YA, Choi JJ, et al. The expression of the miRNA-200 family in endometrial endometrioid carcinoma[J]. Gynecol Oncol, 2011,120(1):56-62. |
| [9] | Ratner ES, Tuck D, Richter C, et al. MicroRNA signatures differentiate uterine cancer tumor subtypes[J]. Gynecol Oncol, 2010,118(3):251-257. |
| [10] | Chung TK, Cheung TH, Huen NY, et al. Dysregulated micro-RNAs and their predicted targets associated with endometrioid endometrial adenocarcinoma in Hong Kong women[J]. Int J Can-cer, 2009,124(6):1358-1365. |
| [11] | 刘霞, 夏伟, 代荫梅, 等. 子宫内膜腺癌组织中miRNA的差异表达[J]. 中华医学杂志, 2009,89(19):1365-1367. |
| [12] | Wu W, Lin Z, Zhuang Z, et al. Expression profile of mammalian microRNAs in endometrioid adenocarcinoma[J]. Eur J Cancer Prev, 2009,18(1):50-55. |
| [13] | Hiroki E, Akahira J, Suzuki F, et al. Changes in microRNA expression levels correlate with clinicopathological features and prognoses in endometrial serous adenocarcinomas[J]. Cancer Sci, 2010,101(1):241-249. |
| [14] |
Devor EJ, Hovey AM, Goodheart MJ, et al. microRNA expression profiling of endometrial endometrioid adenocarcinomas and serous adenocarcinomas reveals profiles containing shared, unique and differentiating groups of microRNAs[J]. Oncol Rep, 2011,26(4):995-1002.
pmid: 21725615 |
| [15] |
Snowdon J, Zhang X, Childs T, et al. The microRNA-200 family is upregulated in endometrial carcinoma[J]. PLoS One, 2011,6(8):e22828.
doi: 10.1371/journal.pone.0022828 pmid: 21897839 |
| [16] |
Torres A, Torres K, Pesci A, et al. Diagnostic and prognostic significance of miRNA signatures in tissues and plasma of endome-trioid endometrial carcinoma patients[J]. Int J Cancer, 2013,132(7):1633-1645.
doi: 10.1002/ijc.27840 pmid: 22987275 |
| [17] |
Bauer KM, Hummon AB. Effects of the miR-143/-145 microRNA cluster on the colon cancer proteome and transcriptome[J]. J Proteome Res, 2012,11(9):4744-4754.
doi: 10.1021/pr300600r |
| [18] |
Volinia S, Calin GA, Liu CG, et al. A microRNA expression signature of human solid tumors defines cancer gene targets[J]. Proc Natl Acad Sci USA, 2006,103(7):2257-2261.
doi: 10.1073/pnas.0510565103 pmid: 16461460 |
| [19] |
Zhang X, Dong Y, Ti H, et al. Down-regulation of miR-145 and miR-143 might be associated with DNA methyltransferase 3B overexpression and worse prognosis in endometrioid carcinomas[J]. Hum Pathol, 2013,44(11):2571-2580.
doi: 10.1016/j.humpath.2013.07.002 |
| [1] | Hao FU, Luyan SHEN, Bingyang HUANG, Shaohua MA. Clinical strategies for perioperative management of esophageal squamous cell carcinoma in the immunotherapy era [J]. Journal of Peking University (Health Sciences), 2026, 58(2): 266-271. |
| [2] | Haoming YIN, Zijie WANG, Fan SHU, Zhanyi ZHANG, Hui LIANG, Shudong ZHANG. Expression and significance of the FABP6 long transcript in clear cell renal cell carcinoma [J]. Journal of Peking University (Health Sciences), 2026, 58(2): 393-398. |
| [3] | Jialin LI, Liqiao CHEN, Jiatian TANG, Yan WU, Anqiang WANG. Conversion therapy for hepatoid adenocarcinoma of the stomach: A case report [J]. Journal of Peking University (Health Sciences), 2026, 58(2): 399-404. |
| [4] | Zichen PAN, Kai CHEN, Yukun HOU, Bohan YANG, Jixin ZHANG, Yongsu MA, Xiaodong TIAN, Yinmo YANG. Research progress in clinical pathology and molecular mechanisms of pancreatic adenosquamous carcinoma [J]. Journal of Peking University (Health Sciences), 2026, 58(2): 431-435. |
| [5] | Xiaolin WANG, Luyao LI, Wen ZHANG, Hongyan WANG. Clinicopathological analysis of mesonephric-like adenocarcinoma in the corpusuteri: A report of 3 cases [J]. Journal of Peking University (Health Sciences), 2025, 57(6): 1208-1212. |
| [6] | Weihua HOU, Shujie SONG, Zhongyue SHI, Lu LIU, Mulan JIN. Neuroendocrine carcinoma with significantly vacuolar nucleus at the esophagogastric junction: A case report [J]. Journal of Peking University (Health Sciences), 2025, 57(5): 1005-1009. |
| [7] | Shang XIE, Luming WANG, Xinyuan ZHANG, Qiushi FENG, Yangyang XIA, Ziwei DAI, Xiaofeng SHAN, Zhigang CAI. Construction and application of oral squamous cell carcinoma organoid bank [J]. Journal of Peking University (Health Sciences), 2025, 57(5): 847-851. |
| [8] | Shaohai TANG, Baoming YANG, Jiankun LI, Lili ZHAO, Yifan WANG, Shunxiang WANG. HDAC2-mediated H3K27 acetylation promotes the proliferation and migration of hepatocellular carcinoma cells [J]. Journal of Peking University (Health Sciences), 2025, 57(5): 884-894. |
| [9] | Xiaoyong YANG, Fan ZHANG, Lulin MA, Cheng LIU. Clinical characteristics and influencing factors of extraglandular invasion of prostatic ductal adenocarcinoma [J]. Journal of Peking University (Health Sciences), 2025, 57(5): 956-960. |
| [10] | Shenmo LI, Dandan SU, Jiyu LIN, Haodong SONG, Lulin MA, Xiaofei HOU, Guoliang WANG, Hongxian ZHANG, Jianfei YE, Shudong ZHANG. Prognostic analysis of laparoscopic simultaneous radical cystectomy and nephroureterectomy [J]. Journal of Peking University (Health Sciences), 2025, 57(5): 961-966. |
| [11] | Boda GUO, Min LU, Guoliang WANG, Hongxian ZHANG, Lei LIU, Xiaofei HOU, Lei ZHAO, Xiaojun TIAN, Shudong ZHANG. Clinicopathological and prognostic differences between clear cell and non-clear cell renal cell carcinoma with venous tumor thrombus [J]. Journal of Peking University (Health Sciences), 2025, 57(4): 644-649. |
| [12] | Zhanyi ZHANG, Min LU, Yuehao SUN, Jinghan DONG, Xiaofei HOU, Chunlei XIAO, Guoliang WANG, Xiaojun TIAN, Lulin MA, Hongxian ZHANG, Shudong ZHANG. Clinicopathological features and survival analysis of TFE3-rearranged renal cell carcinoma with venous tumor thrombus [J]. Journal of Peking University (Health Sciences), 2025, 57(4): 650-661. |
| [13] | Zezhen ZHOU, Liyuan GE, Fan ZHANG, Shaohui DENG, Ye YAN, Hongxian ZHANG, Guoliang WANG, Lei LIU, Yi HUANG, Shudong ZHANG. A retrospective matching study of partial nephrectomy and radical nephrectomy for pathological T3a stage renal cell carcinoma [J]. Journal of Peking University (Health Sciences), 2025, 57(4): 704-710. |
| [14] | Li XU, Wen SHI, Yuehua LI, Yajun SHEN, Shang XIE, Xiaofeng SHAN, Zhigang CAI. LIM and calponin homology domains 1 may function as promising biological markers to aid in the prognostic prediction of oral squamous cell carcinoma [J]. Journal of Peking University (Health Sciences), 2025, 57(1): 19-25. |
| [15] | Minying MA, Xiaoqin CHAO, Yang ZHAO, Guoting ZHAO. Effects of LncRNA SNHG20 on epithelial mesenchymal transition and microtubule formation in human oral squamous cell carcinoma cells through targeted regulation of the miR-520c-3p/RAB22A pathway [J]. Journal of Peking University (Health Sciences), 2025, 57(1): 26-32. |
|
||