Journal of Peking University (Health Sciences) ›› 2026, Vol. 58 ›› Issue (2): 342-350. doi: 10.19723/j.issn.1671-167X.2026.02.019
Previous Articles Next Articles
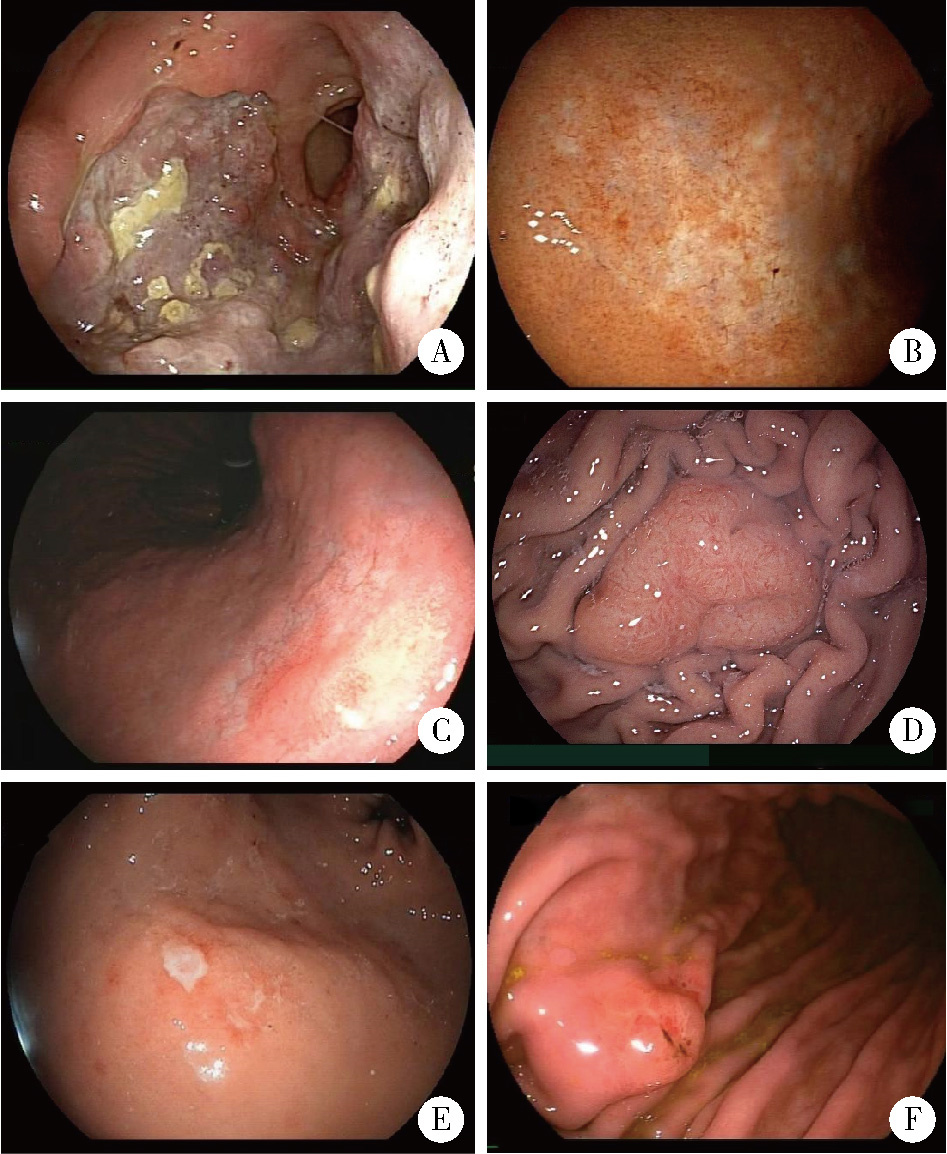
Endoscopic characteristics of primary gastric lymphoma and prediction of treatment response
Jingyao WEI1, Juxiang YE2, Meiling ZHOU1, Weiwei FU1, Xin LIU1, Kangle ZHAI1, Yanyan SHI3, Shigang DING1,*( ), Jing ZHANG1,*(
), Jing ZHANG1,*( )
)
- 1. Department of Gastroenterology, Peking University Third Hospital, Beijing 100191, China
2. Department of Pathology, Peking University Third Hospital, Beijing 100191, China
3. Clinical Epidemiology Research Center, Peking University Third Hospital, Beijing 100191, China
CLC Number:
- R735.2
| 1 |
doi: 10.1097/MEG.0000000000001651 |
| 2 |
National Comprehensive Cancer Network (NCCN). NCCN Guidelines Navigator: B-cell lymphomas. Version 3.2025[EB/OL]. [2025-10-01]. https://www.nccn.org/guidelines/guidelines-detail?category=1&id=1480.
|
| 3 |
doi: 10.1016/j.annonc.2025.07.014 |
| 4 |
doi: 10.3748/wjg.v17.i6.697 |
| 5 |
doi: 10.1093/oxfordjournals.annonc.a058869 |
| 6 |
doi: 10.1038/s41375-022-01620-2 |
| 7 |
doi: 10.1111/bjh.12078 |
| 8 |
doi: 10.1186/s12957-019-1659-8 |
| 9 |
Herlevic V, Reynolds SB, Morris JD. Gastric Lymphoma[M/OL]. In: StatPearls[Internet]. Treasure Island (FL): StatPearls Publishing, 2024[2025-10-01]. https://www.ncbi.nlm.nih.gov/books/NBK567799/.
|
| 10 |
doi: 10.3390/diagnostics14060607 |
| 11 |
doi: 10.3390/cancers15153811 |
| 12 |
doi: 10.1186/s12876-024-03476-5 |
| 13 |
doi: 10.1111/bjh.19064 |
| 14 |
doi: 10.1055/a-2685-2913 |
| 15 |
doi: 10.1111/bjh.19470 |
| 16 |
doi: 10.12998/wjcc.v10.i33.12447 |
| 17 |
doi: 10.1309/AJCPZQ1GXBBNG8OG |
| 18 |
doi: 10.1038/s41598-024-55663-9 |
| 19 |
doi: 10.1038/s41598-020-71792-3 |
| 20 |
doi: 10.1053/gast.2002.33047 |
| 21 |
doi: 10.1182/blood-2005-01-0004 |
| 22 |
doi: 10.3324/haematol.2019.228775 |
| 23 |
doi: 10.3390/cancers14020446 |
| 24 |
doi: 10.1007/s00277-023-05130-8 |
| 25 |
doi: 10.1016/j.ijrobp.2024.10.020 |
| [1] | Jiafu JI, Jingtao WEI, Ke JI, Zhaode BU. Bottlenecks and breakthroughs in gastric cancer diagnosis and treatment: Towards a new era of precision and intelligent integration [J]. Journal of Peking University (Health Sciences), 2026, 58(2): 231-238. |
| [2] | Zhaode BU, Mengyu FENG, Ke JI. Practice and reflection on sentinel lymph node navigation surgery for early gastric cancer [J]. Journal of Peking University (Health Sciences), 2026, 58(2): 239-243. |
| [3] | Bixian LUO, Hongming LIU, Weixun XIE, Weihua GONG. Novel clinical insights and frontier issues in alpha- fetoprotein-producing gastric cancer [J]. Journal of Peking University (Health Sciences), 2026, 58(2): 257-265. |
| [4] | Youdong LIU, Yajun LYU, Jie CHEN, Mingde ZANG, Hongda PAN, Xiaowen LIU, Jun LU, Fenglin LIU. Clinical efficacy and safety of totally laparoscopic subtotal gastrectomy with cardia-gastric fundus preservation in middle-upper gastric cancer [J]. Journal of Peking University (Health Sciences), 2026, 58(2): 301-306. |
| [5] | Jialin LI, Liqiao CHEN, Jiatian TANG, Yan WU, Anqiang WANG. Conversion therapy for hepatoid adenocarcinoma of the stomach: A case report [J]. Journal of Peking University (Health Sciences), 2026, 58(2): 399-404. |
| [6] | 郭芷均, 俎明, 马超, 张贺军, 张静, 丁士刚. [J]. Journal of Peking University (Health Sciences), 2026, 58(2): 410-415. |
| [7] | Bin LI, Han LIANG. Robotic gastrectomy: Research progress and practical challenges [J]. Journal of Peking University (Health Sciences), 2026, 58(2): 416-422. |
| [8] | Weihua HOU, Shujie SONG, Zhongyue SHI, Lu LIU, Mulan JIN. Neuroendocrine carcinoma with significantly vacuolar nucleus at the esophagogastric junction: A case report [J]. Journal of Peking University (Health Sciences), 2025, 57(5): 1005-1009. |
| [9] | Zhemin LI, Jiafu JI, Guoxin LI, Ziyu LI, Zhaode BU, Xiangyu GAO, Di DONG, Lei TANG, Xiaofang XING, Shuqin JIA, Ting GUO, Lianhai ZHANG, Fei SHAN, Xin JI, Anqiang WANG. Development and dissemination of precision medicine approaches in gastric cancer management [J]. Journal of Peking University (Health Sciences), 2025, 57(5): 864-867. |
| [10] | Wei-hua HOU,Shu-jie SONG,Zhong-yue SHI,Mu-lan JIN. Clinicopathological features of Helicobacter pylori-negative early gastric cancer [J]. Journal of Peking University (Health Sciences), 2023, 55(2): 292-298. |
| [11] | Ju-mei LIU,Li LIANG,Ji-xin ZHANG,Long RONG,Zi-yi ZHANG,You WU,Xu-dong ZHAO,Ting LI. Pathological evaluation of endoscopic submucosal dissection for early gastric cancer and precancerous lesion in 411 cases [J]. Journal of Peking University (Health Sciences), 2023, 55(2): 299-307. |
| [12] | NIU Zhan-yue,XUE Yan,ZHANG Jing,ZHANG He-jun,DING Shi-gang. Analysis of endoscopic and pathological features of gastric adenomatous polyps and risk factors for canceration [J]. Journal of Peking University (Health Sciences), 2021, 53(6): 1122-1127. |
| [13] | Ying-chao WU,Yun-long CAI,Long RONG,Ji-xin ZHANG,Jin LIU,Xin WANG. Characteristics of lymph node metastasis and evaluating the efficacy of endoscopic submucosal dissection in early gastric cancer [J]. Journal of Peking University (Health Sciences), 2020, 52(6): 1093-1097. |
| [14] | Yang YANG,Yi-qiang LIU,Xiao-hong WANG,Ke JI,Zhong-wu LI,Jian BAI,Ai-rong YANG,Ying HU,Hai-bo HAN,Zi-yu LI,Zhao-de BU,Xiao-jiang WU,Lian-hai ZHANG,Jia-fu JI. Clinicopathological and molecular characteristics of Epstein-Barr virus associated gastric cancer: a single center large sample case investigation [J]. Journal of Peking University(Health Sciences), 2019, 51(3): 451-458. |
| [15] | Xin LIU,Jing ZHANG,Ye WANG,He-jun ZHANG,Shi-gang DING,Li-ya Zhou. Characteristics analysis of early gastric cancer under white light endoscopy [J]. Journal of Peking University(Health Sciences), 2019, 51(2): 302-306. |
|
||