北京大学学报(医学版) ›› 2026, Vol. 58 ›› Issue (2): 399-404. doi: 10.19723/j.issn.1671-167X.2026.02.027
胃肝样腺癌转化治疗1例
李嘉临1, 陈力侨1, 唐家天1, 吴艳2, 王安强1,*( )
)
- 1. 北京大学肿瘤医院暨北京市肿瘤防治研究所胃肠肿瘤中心,北京 100142
2. 北京大学肿瘤医院暨北京市肿瘤防治研究所病理科,恶性肿瘤发病机制及转化研究教育部重点实验室,北京 100142
Conversion therapy for hepatoid adenocarcinoma of the stomach: A case report
Jialin LI1, Liqiao CHEN1, Jiatian TANG1, Yan WU2, Anqiang WANG1,*( )
)
- 1. Key laboratory of Carcinogenesis and Translational Research (Ministry of Education), Center of Gastrointestinal Cancer, Peking University Cancer Hospital & Institute, Beijing 100142, China
2. Key laboratory of Carcinogenesis and Translational Research (Ministry of Education), Department of Pathology, Peking University Cancer Hospital & Institute, Beijing 100142, China
摘要:
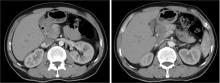
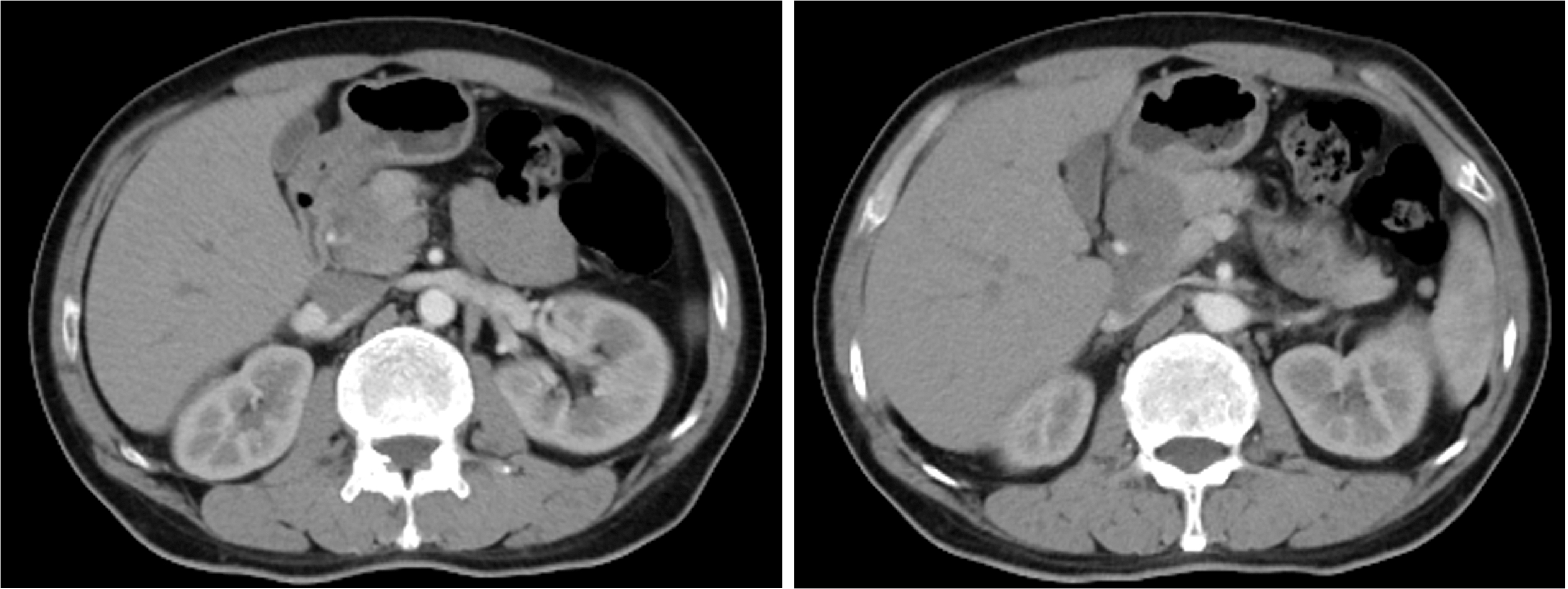
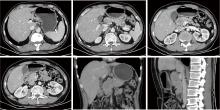
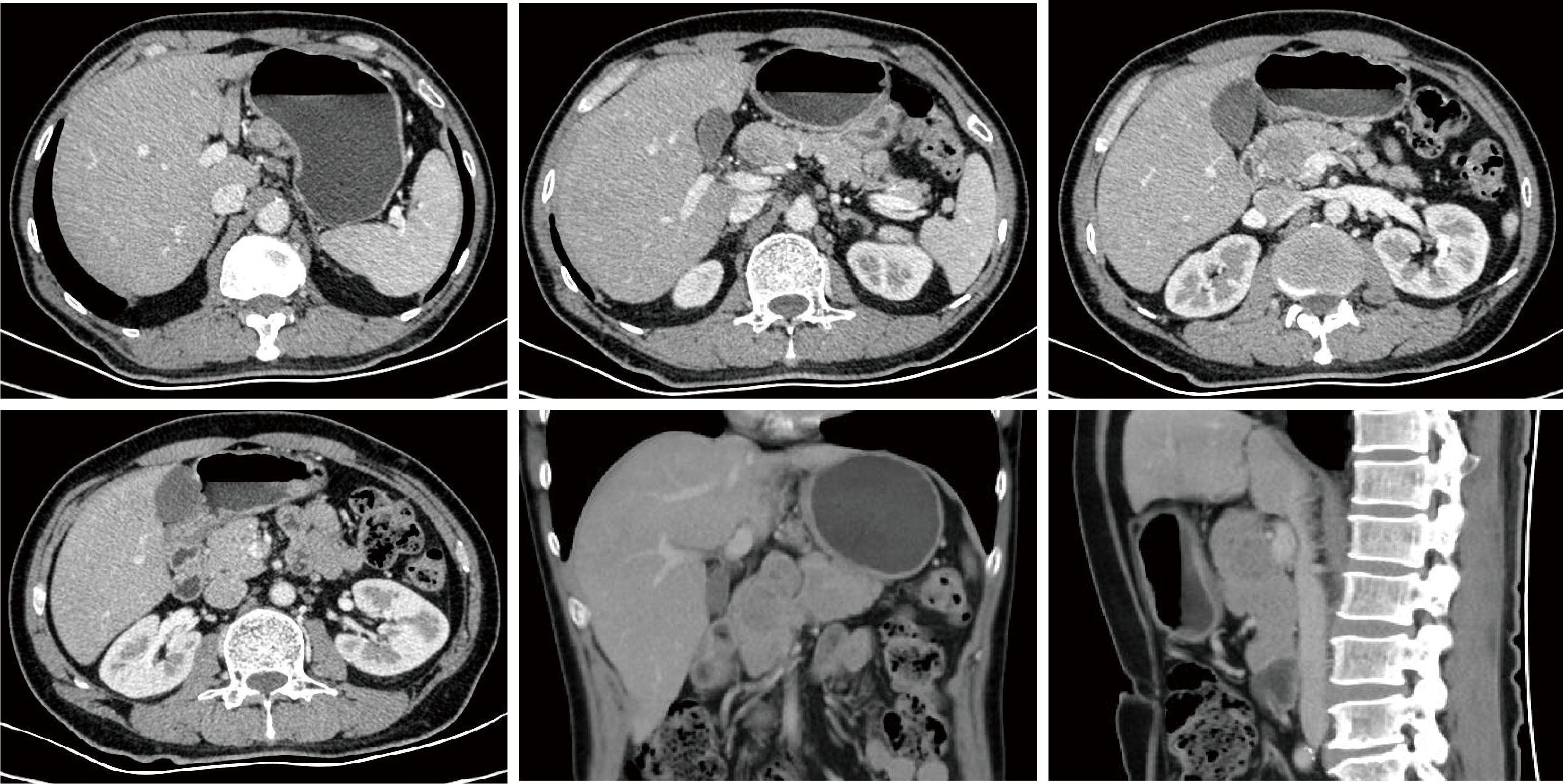
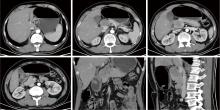
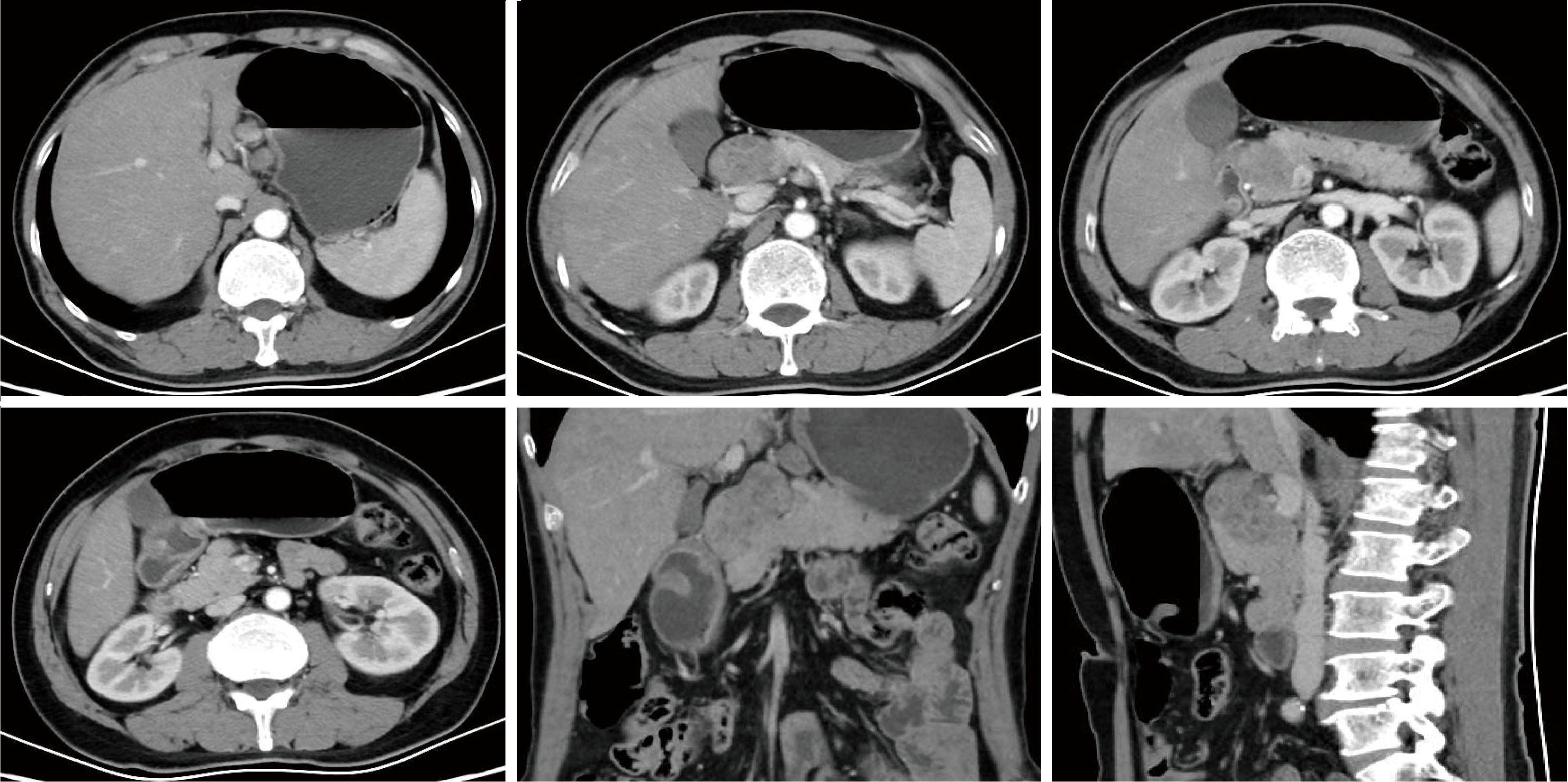
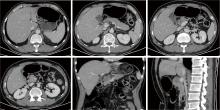
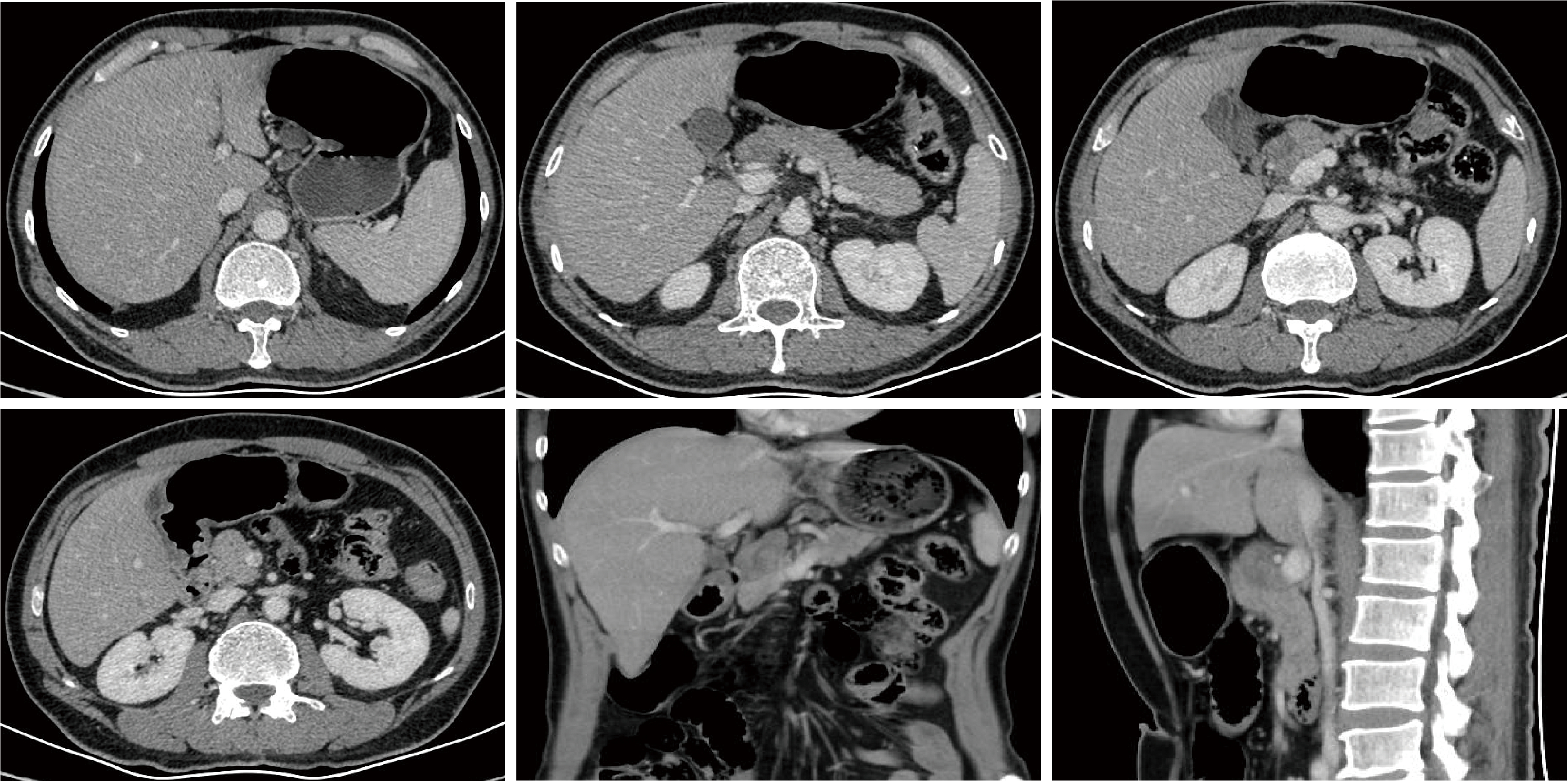
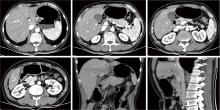
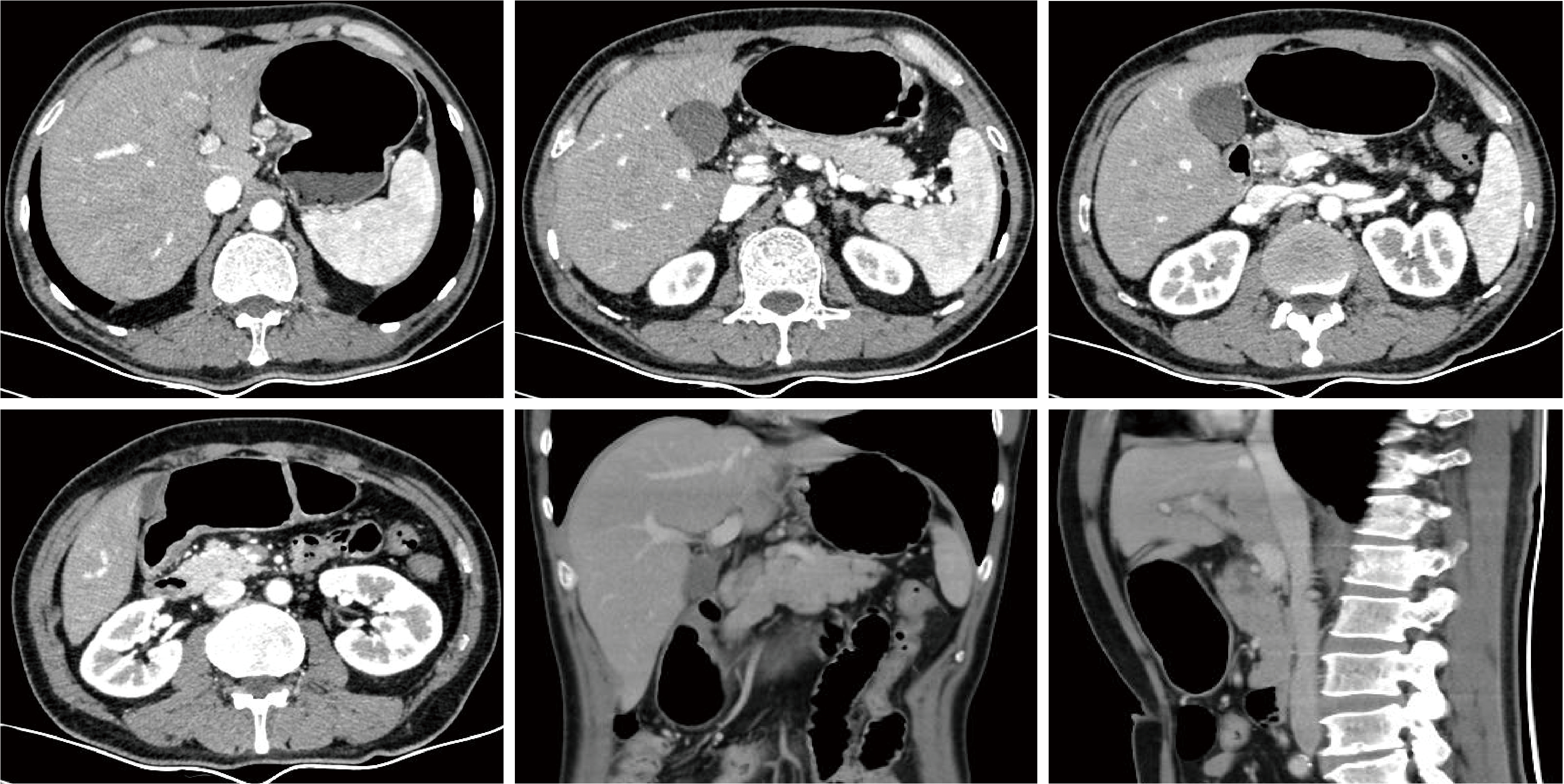
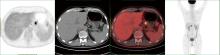
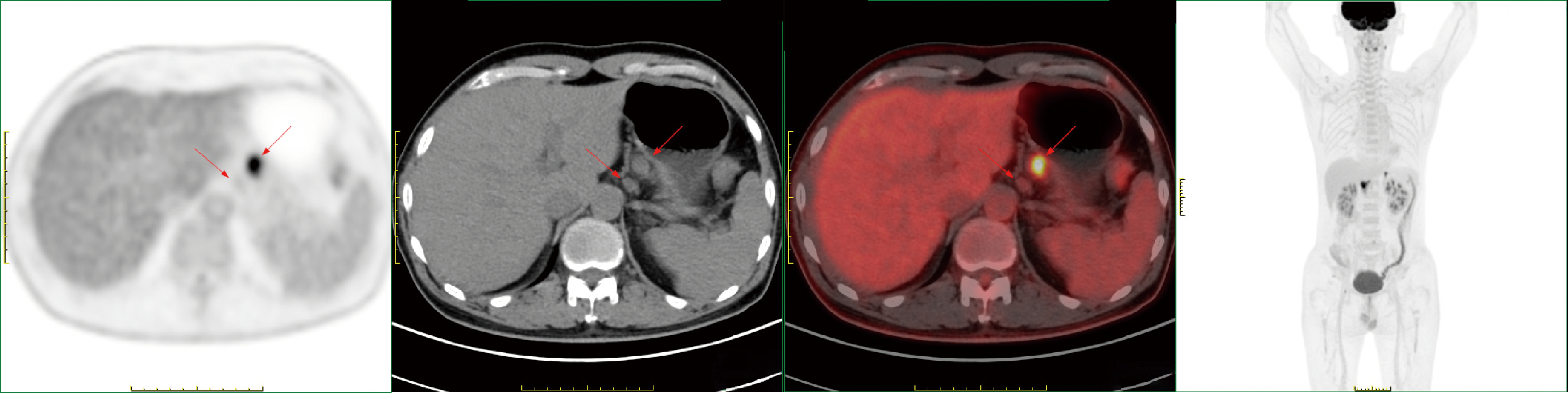
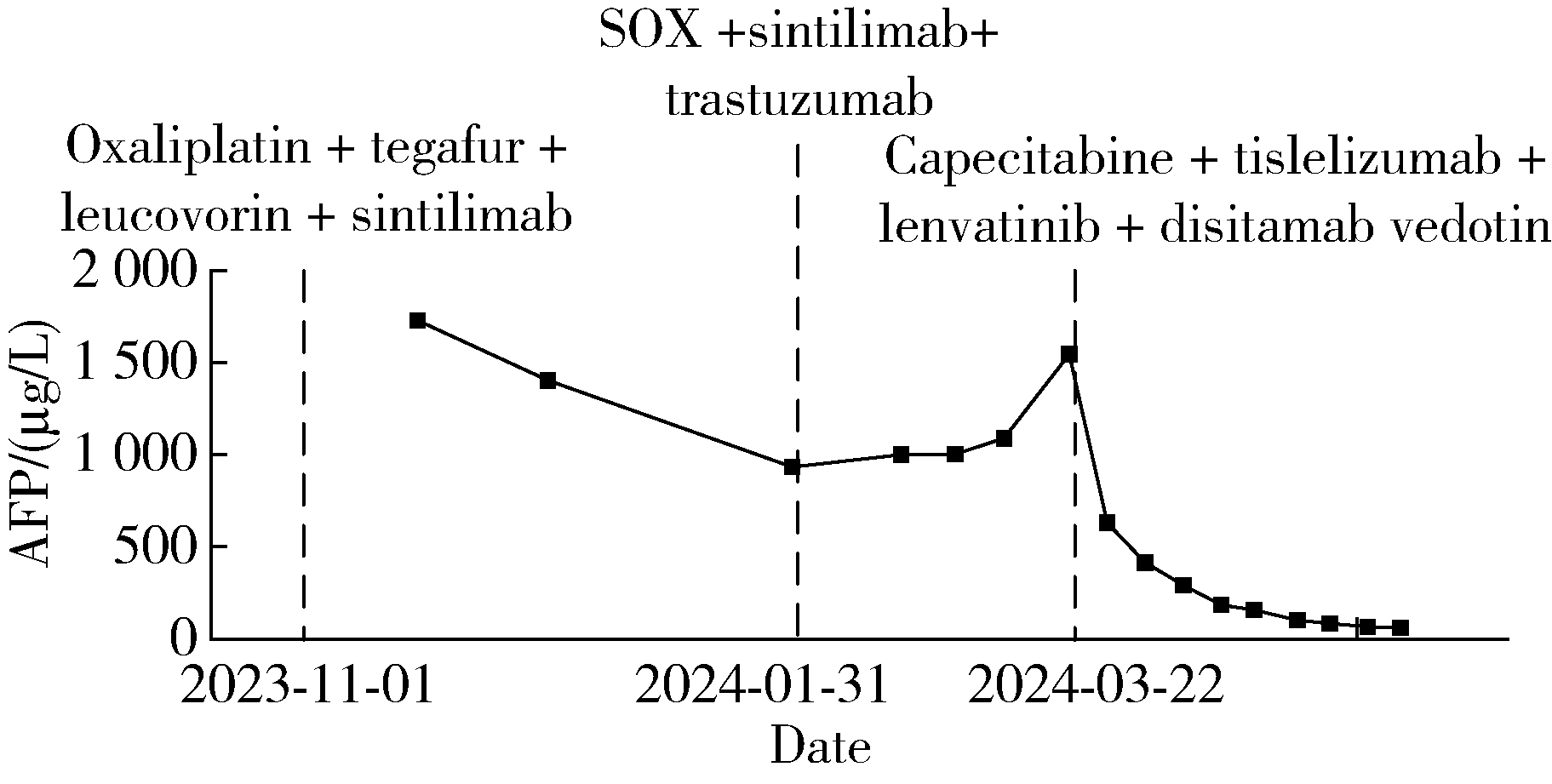
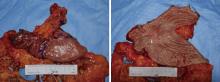
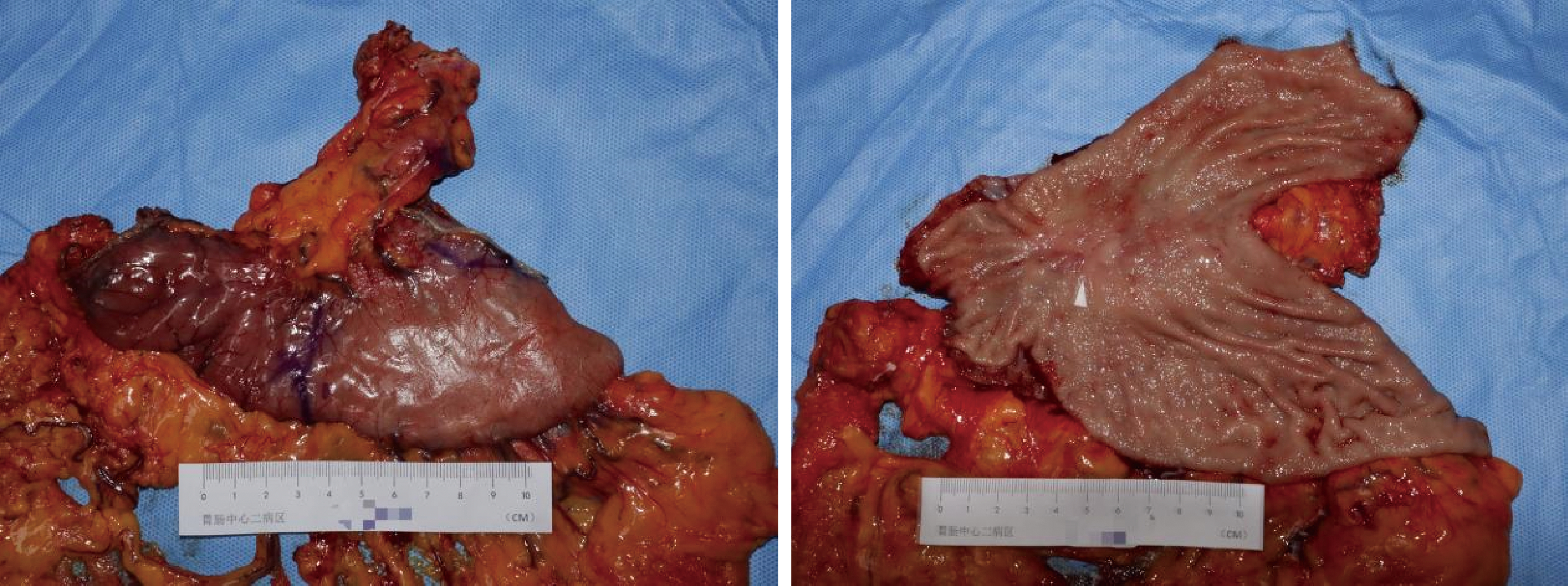
胃肝样腺癌(hepatoid adenocarcinoma of the stomach,HAS)是一种罕见且高度侵袭性的胃癌亚型,其组织学特征类似于肝细胞癌,并常伴有血清甲胎蛋白(alpha-fetoprotein,AFP)显著升高。HAS预后极差,对于初始不可切除的HAS,目前尚无标准治疗方案,因此,转化治疗成为实现根治的关键策略。本文报道1例56岁男性患者,初诊为局部晚期、初始不可切除的HAS,伴有血清AFP升高。一线转化治疗采用奥沙利铂为基础的化疗联合信迪利单抗,后续加用曲妥珠单抗,但治疗失败,疾病进展。肿瘤组织检测显示人表皮生长因子受体2(human epidermal growth factor receptor 2,HER2)扩增。随后启动二线高度个体化的联合方案,包括维迪西妥单抗(靶向HER2的抗体药物偶联物)、仑伐替尼(多靶点酪氨酸激酶抑制剂)、替雷利珠单抗及短期应用的卡培他滨。该方案持续降低患者血清AFP水平,并使原发肿瘤及转移淋巴结明显缩小,达到部分缓解,成功实现降期。随后患者成功接受R0根治性远端胃切除术,术后病理证实为ypT1bN1期。术后继续给予2个周期相同靶向-免疫维持治疗,后因累积毒性转为观察随访。术后1年半随访,患者无肿瘤复发。本病例表明,对于化疗耐药、HER2阳性的HAS,采用以HER2靶向抗体药物偶联物为核心,联合抗血管生成药物与免疫治疗的强效、分子导向策略,可有效克服治疗耐药,实现显著降期,并获得长期无病生存。
中图分类号:
- R735.2
| 1 |
doi: 10.1002/cac2.12336 |
| 2 |
doi: 10.1038/s41388-021-01976-2 |
| 3 |
doi: 10.1158/0008-5472.CAN-07-5962 |
| 4 |
doi: 10.1007/s10549-015-3503-3 |
| 5 |
doi: 10.1002/cac2.12214 |
| 6 |
doi: 10.1007/s40265-019-01116-x |
| 7 |
doi: 10.1158/1078-0432.CCR-20-2571 |
| 8 |
doi: 10.1186/s12885-022-09405-7 |
| 9 |
doi: 10.1016/S1470-2045(20)30271-0 |
| [1] | 季加孚, 韦静涛, 季科, 步召德. 胃癌诊疗的瓶颈与破局:迈向精准化与智能化融合的新纪元[J]. 北京大学学报(医学版), 2026, 58(2): 231-238. |
| [2] | 高加勒, 张忠涛. 局部进展期直肠癌精准治疗现状与展望[J]. 北京大学学报(医学版), 2026, 58(2): 247-250. |
| [3] | 王海, 江一舟. 靶向血管治疗在乳腺癌精准治疗中的分子机制与临床应用[J]. 北京大学学报(医学版), 2026, 58(2): 251-256. |
| [4] | 刘友东, 吕亚军, 陈杰, 臧明德, 潘宏达, 刘晓文, 陆俊, 刘凤林. 全腹腔镜保留贲门胃底胃次全切除术治疗中上部胃癌的疗效及安全性[J]. 北京大学学报(医学版), 2026, 58(2): 301-306. |
| [5] | 李斌, 梁寒. 机器人胃癌根治术:研究进展与实践挑战[J]. 北京大学学报(医学版), 2026, 58(2): 416-422. |
| [6] | 王晓林, 李璐瑶, 张雯, 王鸿雁. 3例子宫体中肾样腺癌的临床病理学分析[J]. 北京大学学报(医学版), 2025, 57(6): 1208-1212. |
| [7] | 侯卫华, 宋书杰, 石中月, 刘露, 金木兰. 食管胃结合部具有显著空泡状核特征的神经内分泌癌1例[J]. 北京大学学报(医学版), 2025, 57(5): 1005-1009. |
| [8] | 杨小勇, 张帆, 马潞林, 刘承. 前列腺导管腺癌临床特征及腺外侵犯的影响因素[J]. 北京大学学报(医学版), 2025, 57(5): 956-960. |
| [9] | 刘杰, 马茗微, 王庆安, 石明, 尹金鹏, 王占平, 申静涛, 高献书. 基于锥形束CT的前列腺癌放射治疗两种体位固定方式摆位误差比较[J]. 北京大学学报(医学版), 2025, 57(4): 692-697. |
| [10] | 刘苗雨, 王思源, 裴林, 王殊. 同步应用戈舍瑞林对年轻乳腺癌患者新辅助化疗疗效的影响[J]. 北京大学学报(医学版), 2025, 57(2): 291-297. |
| [11] | 罗丹, 黄海建, 陈新, 陈小岩. 原发子宫肝样腺癌2例临床病理分析及文献复习[J]. 北京大学学报(医学版), 2024, 56(6): 1126-1131. |
| [12] | 黄教悌,胡菁,韩博. 治疗相关神经内分泌前列腺癌机制研究与靶向治疗新进展[J]. 北京大学学报(医学版), 2024, 56(4): 557-561. |
| [13] | 邢念增,王明帅,杨飞亚,尹路,韩苏军. 前列腺免活检创新理念的临床实践及其应用前景[J]. 北京大学学报(医学版), 2024, 56(4): 565-566. |
| [14] | 颜野,李小龙,夏海缀,朱学华,张羽婷,张帆,刘可,刘承,马潞林. 前列腺癌根治术后远期膀胱过度活动症的危险因素[J]. 北京大学学报(医学版), 2024, 56(4): 589-593. |
| [15] | 于书慧,韩佳凝,钟丽君,陈聪语,肖云翔,黄燕波,杨洋,车新艳. 术前盆底肌电生理参数对前列腺癌根治性切除术后早期尿失禁的预测价值[J]. 北京大学学报(医学版), 2024, 56(4): 594-599. |
|
||