北京大学学报(医学版) ›› 2019, Vol. 51 ›› Issue (1): 21-27. doi: 10.19723/j.issn.1671-167X.2019.01.005
大鼠后肢长期抗阻训练对慢性咬肌机械痛觉敏感性的影响
- 北京大学口腔医学院·口腔医院,修复科 口颌功能诊疗研究中心 国家口腔疾病临床医学研究中心 口腔数字化医疗技术和材料国家工程实验室 口腔数字医学北京市重点实验室,北京 100081
Effect of long-term resistance exercise on masseter muscle mechanical hyperalgesia in rats
Shu-dong YAN,Guang-ju YANG( ),Si-yi MO,Yun LIU,Qiu-fei XIE(
),Si-yi MO,Yun LIU,Qiu-fei XIE( )
)
- Department of Prosthodontics,Center for Oral and Jaw Functional Diagnosis, Peking University School and Hospital of Stomatology & National Clinical Research Center for Oral Diseases & National Engineering Laboratory for Digital and Material Technology of Stomatology & Beijing Key Laboratory of Digital Stomatology,Beijing 100081, China
摘要:
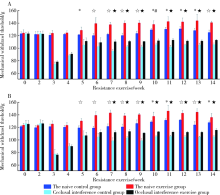
目的:研究长期抗阻训练对大鼠慢性咬肌机械痛觉敏感性的影响,为临床慢性咀嚼疼痛的非药物治疗提供理论依据。方法:选用16只雄性Sprague-Dawley大鼠(220~250 g),随机分为空白对照组、抗阻训练组、慢性咬肌痛组、慢性咬肌痛抗阻训练组4个组,每组4只,慢性咬肌痛大鼠(慢性咬肌痛组和慢性咬肌痛抗阻训练组)采用咬合干扰模型建模,抗阻训练大鼠(抗阻训练组和慢性咬肌痛抗阻训练组)采用食物诱导法进行负重蹲起训练,每次30 min,每日1次,每周训练5 d,总计14周。每天记录大鼠抗阻训练情况,采用改良电子测痛仪每周测定各组大鼠双侧咬肌机械刺激反应阈值,每周记录体重变化。长期抗阻训练结束后,采用自制抓力计测定大鼠后肢抓力,分离后肢肌肉(腓肠肌和比目鱼肌)及腓骨,测定肌肉质量和腓骨长度,计算后肢肌肉质量、肌肉质量/体重比和肌肉质量/腓骨长度比。结果:空白对照组和抗阻训练组第0~4周双侧咬肌机械刺激反应阈值差异无统计学意义(P>0.05), 第5~14周抗阻训练组的阈值较空白对照组高(P<0.05)。慢性咬肌痛组和慢性咬肌痛抗阻训练组在第0~6周双侧咬肌机械刺激反应阈值均先下降后上升,组间阈值差异无统计学意义(P>0.05), 第7~14周慢性咬肌痛抗阻训练组的阈值较慢性咬肌痛组的显著增高(P<0.05)。14周抗阻训练后,非抗阻训练大鼠体重明显大于抗阻训练大鼠[(462±6) g vs.(418±14) g, P<0.05], 大鼠后肢抓力在抗阻训练组明显更高[(6.75±0.13) N vs.(5.41±0.15) N, P<0.01]。结论:长期抗阻训练可降低健康大鼠和慢性咬肌痛觉过敏大鼠双侧咬肌机械痛觉敏感性,为长期抗阻训练临床治疗慢性咀嚼肌痛提供了基础研究证据。
中图分类号:
- R78
| [1] |
Soyannwo OA . Improved neuropathic pain treatment in developing countries-a critical review of WHO essential list[J]. Pain, 2015,156(5):763-764.
doi: 10.1097/j.pain.0000000000000140 pmid: 25719619 |
| [2] |
Cavalieri TA . Management of pain in older adults[J]. J Am Osteopath Assoc, 2005,105(Suppl 3):12-19.
doi: 10.1016/B978-0-323-08340-9.00034-7 pmid: 18154193 |
| [3] |
Dworkin RH, O’Connor AB, Audette J , et al. Recommendations for the pharmacological management of neuropathic pain: an overview and literature update[J]. Mayo Clin Proc, 2010,85(3):S3-S14.
doi: 10.4065/mcp.2009.0649 pmid: 20194146 |
| [4] |
O’Connor AB, Dworkin RH . Treatment of neuropathic pain: an overview of recent guidelines[J]. Am J Med, 2009,122(Suppl 10):S22-S32
doi: 10.1016/j.amjmed.2009.04.007 pmid: 19801049 |
| [5] |
Latham N, Liu C . Strength training in older adults: the benefits for osteoarthritis[J]. Clin Geriatr Med, 2010,26(3):445-459.
doi: 10.1016/j.cger.2010.03.006 pmid: 3606891 |
| [6] |
Jansen M J, Viechtbauer W, Lenssen AF , et al. Strength training alone, exercise therapy alone, and exercise therapy with passive manual mobilisation each reduce pain and disability in people with knee osteoarthritis: a systematic review[J]. J Physiother, 2011,57(1):11-20.
doi: 10.1016/S1836-9553(11)70002-9 pmid: 0034625 |
| [7] |
Strasser B, Leeb G, Strehblow C , et al. The effects of strength and endurance training in patients with rheumatoid arthritis[J]. Clin Rheumatol, 2011,30(5):623-632.
doi: 10.1007/s10067-010-1584-2 pmid: 20931346 |
| [8] |
Harts CC, Helmhout PH, de Bie RA , et al. A high-intensity lumbar extensor strengthening program is little better than a low-intensity program or a waiting list control group for chronic low back pain: a randomised clinical trial[J]. Aust J Physiother, 2008,54(1):23-31.
doi: 10.1016/S0004-9514(08)70062-X pmid: 18298356 |
| [9] |
Kami K, Tajima F, Senba E . Exercise-induced hypoalgesia: potential mechanisms in animal models of neuropathic pain[J]. Anat Sci Int, 2017,92(1):79-90.
doi: 10.1007/s12565-016-0360-z |
| [10] |
Cao Y, Xie QF, Li K , et al. Experimental occlusal interference induces long-term masticatory muscle hyperalgesia in rats[J]. Pain, 2009,144(3):287-293.
doi: 10.1016/j.pain.2009.04.029 pmid: 19473767 |
| [11] |
Cao Y, Li K, Fu KY , et al. Central sensitization and MAPKs are involved in occlusal interference-Induced facial pain in rats[J]. J Pain, 2013,14(8):793-807.
doi: 10.1016/j.jpain.2013.02.005 pmid: 3735867 |
| [12] |
李雪姣, 曹烨, 谢秋菲 , 等. 牙合干扰致大鼠咀嚼肌机械痛觉过敏的特点研究[J]. 中华口腔医学杂志, 2014,49(10):596-599.
doi: 10.3760/cma.j.issn.1002-0098.2014.10.006 |
| [13] |
Klitgaard H . A model for quantitative strength training of hindlimb muscles of the rat[J]. J Appl Physiol, 1988,64(4):1740-1745.
doi: 10.1055/s-2007-1025001 pmid: 3379005 |
| [14] |
Norenberg KM, Fitts RH . Contractile responses of the rat gastrocnemius and soleus muscles to isotonic resistance exercise[J]. J Appl Physiol, 2004,97(6):2322-2332.
doi: 10.1152/japplphysiol.00955.2003 pmid: 15322071 |
| [15] |
De LA, Pierno S, Liantonio A , et al. Enhanced dystrophic progression in mdx mice by exercise and beneficial effects of taurine and insulin-like growth factor-1[J]. J Pharmacol Exp Ther, 2003,304(1):453-463.
doi: 10.1124/jpet.102.041343 pmid: 12490622 |
| [16] |
Jr HT, Farrar RP . Physiological hypertrophy of the FHL muscle following 8 weeks of progressive resistance exercise in the rat[J]. Can J Appl Physiol, 2004,29(1):16-31.
doi: 10.1139/h04-002 pmid: 15001801 |
| [17] |
Alves JP, Nunes RB, Stefani GP , et al. Resistance training improves hemodynamic function, collagen deposition and inflammatory profiles: experimental model of heart failure[J]. PLoS One, 2014,9(10):e110317.
doi: 10.1371/journal.pone.0110317 pmid: 4207701 |
| [18] | Lee S, Farrar RP . Resistance training induces muscle-specific changes in muscle mass and function in rat[J]. J Exerc Physiol Online, 2003,6(2):80-87. |
| [19] |
Safarzade A, Talebigarakani E . Short term resistance training enhanced plasma apoA-I and FABP4 levels in streptozotocin-induced diabetic rats[J]. J Diabetes Metab Disord, 2014,13(1):41.
doi: 10.1186/2251-6581-13-41 pmid: 24593955 |
| [20] |
Gerrits MM, Van Oppen P, Leone SS , et al. Pain, not chronic disease, is associated with the recurrence of depressive and anxiety disorders[J]. BMC psychiatry, 2014,14(1):187-198.
doi: 10.1186/1471-244X-14-187 pmid: 24965597 |
| [21] |
Yazdi M, Yilmaz Z, Renton T , et al. Psychological morbidity in chronic orofacial pain and headaches[J]. Oral Surgery, 2012,5(4):173-181.
doi: 10.1111/ors.12000 |
| [22] |
Weisberg JN, Boatwright BA . Mood, anxiety and personality traits and states in chronic pain[J]. Pain, 2007,133(1/2/3):1-2.
doi: 10.1016/j.pain.2007.10.005 pmid: 17967507 |
| [23] |
Tamaki T, Uchiyama S, Nakano S . A weight-lifting exercise model for inducing hypertrophy in the hindlimb muscles of rats[J]. Med Sci Sports Exerc, 1992,24(8):881-886.
doi: 10.1249/00005768-199208000-00009 pmid: 1406173 |
| [24] |
Legerlotz K, Schjerling P, Langberg H , et al. The effect of running, strength, and vibration strength training on the mechanical, morphological, and biochemical properties of the Achilles tendon in rats[J]. J Appl Physiol, 2007,102(2):564-572.
doi: 10.1152/japplphysiol.00767.2006 pmid: 17038489 |
| [25] |
刘存瑞, 徐啸翔, 曹烨 , 等. 咬合干扰时间因素对大鼠咀嚼肌机械痛觉敏感的影响[J]. 北京大学学报(医学版), 2016,48(1):51-56.
doi: 10.3969/j.issn.1671-167X.2016.01.009 |
| [26] |
曹烨, 李锴, 傅开元 , 等. 咬合干扰致大鼠咬肌组织蛋白基因产物及P物质表达变化[J]. 北京大学学报(医学版), 2010,42(1):50-55.
doi: 10.3969/j.issn.1671-167X.2010.01.012 |
| [27] |
Merrill RL . Central mechanisms of orofacial pain[J]. Dent Clin North Am, 2007,51(1):45-59.
doi: 10.1016/j.cden.2006.09.010 pmid: 17185059 |
| [28] | 刘念, 臧凯凯, 张玉秋 . 外周神经损伤小鼠不同脊髓节段小胶质细胞和星形胶质细胞的激活状态[J]. 生理学报, 2015,67(6):571-582. |
| [29] |
高永静, 纪如荣 . 星形胶质细胞调节慢性疼痛的分子机制[J]. 中国疼痛医学杂志, 2013,19(9):545-552.
doi: 10.3969/j.issn.1006-9852.2013.09.009 |
| [30] |
Piao ZG, Cho IH, Park CK , et al. Activation of glia and microglial p38 MAPK in medullary dorsal horn contributes to tactile hypersensitivity following trigeminal sensory nerve injury[J]. Pain, 2006,121(3):219-231.
doi: 10.1016/j.pain.2005.12.023 pmid: 16495005 |
| [31] |
Kuphal KE, Fibuch EE, Taylor BK . Extended swimming exercise reduces inflammatory and peripheral neuropathic pain in rodents[J]. J Pain, 2007,8(12):989-997.
doi: 10.1016/j.jpain.2007.08.001 pmid: 17890162 |
| [32] |
Shen J, Fox LE, Cheng J . Swim therapy reduces mechanical allodynia and thermal hyperalgesia induced by chronic constriction nerve injury in rats[J]. Pain Med, 2013,14(4):516-525.
doi: 10.1111/pme.12057 pmid: 3625453 |
| [33] |
Koltyn KF, Brellenthin AG, Cook DB , et al. Mechanisms of exercise-induced hypoalgesia.[J]. J Pain, 2014,15(12):1294-1304.
doi: 10.1016/j.jpain.2014.09.006 pmid: 4302052 |
| [34] |
Chen YW, Li YT, Chen YC , et al. Exercise training attenuates neuropathic pain and cytokine expression after chronic constriction injury of rat sciatic nerve.[J]. Anesth Analg, 2012,114(6):1330-1337.
doi: 10.1213/ANE.0b013e31824c4ed4 pmid: 22415536 |
| [35] |
Bobinski F, Martins DF, Bratti T , et al. Neuroprotective and neuroregenerative effects of low-intensity aerobic exercise on sciatic nerve crush injury in mice.[J]. Neuroscience, 2011,194:337-348.
doi: 10.1016/j.neuroscience.2011.07.075 pmid: 21864654 |
| [36] | Katsuya K, Taguchi MS, Fumihiro T , et al. Improvements in impaired GABA and GAD65/67 production in the spinal dorsal horn contribute to exercise-induced hypoalgesia in a mouse model of neuropathic pain [J/OL]. Mol Pain, 2016, 12[2018-10-01]. |
| [37] |
Millan MJ . Descending control of pain[J]. Prog Neurobiol, 2002,66(6):355-474.
doi: 10.1016/S0301-0082(02)00009-6 |
| [38] |
Bobinski F, Taa F, Córdova MM , et al. Role of brainstem serotonin in analgesia produced by low-intensity exercise on neuropathic pain after sciatic nerve injury in mice[J]. Pain, 2015,156(12):2595-2606.
doi: 10.1097/j.pain.0000000000000372 pmid: 26447701 |
| [1] | 李文博, 沈玉凤, 杨咏涛, 单珅瑶, 高梓翔, 温奥楠, 商相宜, 田淯文, 郭殊玮, 王艺蓁, 王勇, 赵一姣. 单侧磨牙咬合干扰的口颌肌表面肌电指标体系构建与判别模型验证[J]. 北京大学学报(医学版), 2026, 58(1): 89-98. |
| [2] | 范莹莹,刘云,曹烨,谢秋菲. 海马参与雌激素加重咬合干扰致去卵巢大鼠慢性咬肌痛敏[J]. 北京大学学报(医学版), 2022, 54(1): 40-47. |
|
||