Journal of Peking University (Health Sciences) ›› 2026, Vol. 58 ›› Issue (2): 423-430. doi: 10.19723/j.issn.1671-167X.2026.02.031
Previous Articles Next Articles
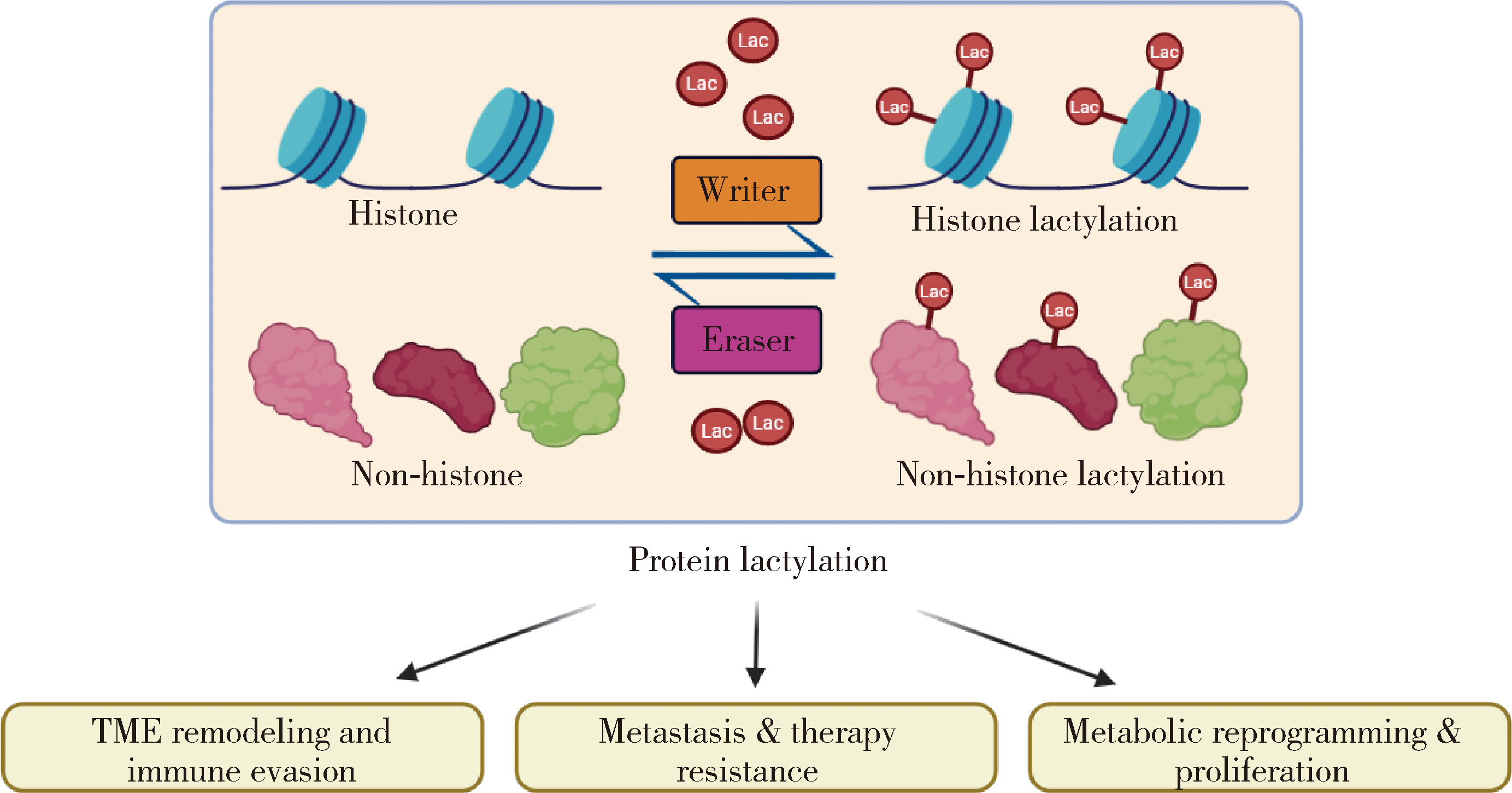
Research progress on protein lactylation modification in malignant tumors
Haifeng DONG, Hengxing CHEN, Changhua ZHANG*( )
)
- Digestive Disease Center, The Seventh Affiliated Hospital, Sun Yat-sen University; Guangdong Provincial Key Laboratory of Digestive Cancer Research, Shenzhen 518000, Guangdong, China
CLC Number:
- R730.2
| 1 |
doi: 10.1152/japplphysiol.00497.2022 |
| 2 |
doi: 10.1016/j.tem.2022.07.004 |
| 3 |
doi: 10.1038/s41392-022-01151-3 |
| 4 |
doi: 10.1016/j.redox.2020.101454 |
| 5 |
doi: 10.1002/cac2.70054 |
| 6 |
doi: 10.1016/j.ebiom.2015.05.022 |
| 7 |
|
| 8 |
doi: 10.3389/fonc.2019.01143 |
| 9 |
doi: 10.1038/s41586-019-1678-1 |
| 10 |
doi: 10.1007/s13238-021-00846-7 |
| 11 |
doi: 10.1038/s41392-024-01918-w |
| 12 |
doi: 10.3389/fimmu.2023.1229397 |
| 13 |
doi: 10.1155/2021/6635225 |
| 14 |
doi: 10.1186/s13046-022-02338-w |
| 15 |
doi: 10.1038/s41592-022-01523-1 |
| 16 |
doi: 10.1002/pmic.202200437 |
| 17 |
doi: 10.1002/pmic.202300003 |
| 18 |
doi: 10.1002/pmic.202200432 |
| 19 |
|
| 20 |
doi: 10.1016/j.isci.2022.104630 |
| 21 |
doi: 10.1016/j.csbj.2021.08.013 |
| 22 |
doi: 10.1016/j.tibs.2019.12.004 |
| 23 |
doi: 10.1016/j.jhazmat.2023.132582 |
| 24 |
doi: 10.1038/s42255-020-0267-9 |
| 25 |
doi: 10.3390/biotech12020038 |
| 26 |
doi: 10.1007/s10571-023-01335-7 |
| 27 |
doi: 10.1016/j.cell.2024.04.002 |
| 28 |
doi: 10.1038/s41586-024-07620-9 |
| 29 |
doi: 10.1016/j.cell.2023.11.022 |
| 30 |
doi: 10.1021/acs.chemrev.7b00181 |
| 31 |
doi: 10.1126/sciadv.abi6696 |
| 32 |
|
| 33 |
doi: 10.1093/jn/135.7.1619 |
| 34 |
doi: 10.1098/rsob.200187 |
| 35 |
doi: 10.1016/j.chembiol.2019.11.005 |
| 36 |
doi: 10.1016/j.drup.2025.101274 |
| 37 |
doi: 10.1002/mco2.292 |
| 38 |
doi: 10.1038/s41392-023-01605-2 |
| 39 |
doi: 10.1016/j.immuni.2024.04.006 |
| 40 |
doi: 10.1186/s13046-023-02815-w |
| 41 |
doi: 10.1016/j.celrep.2022.111122 |
| 42 |
|
| 43 |
doi: 10.3389/fcimb.2022.913815 |
| 44 |
doi: 10.1038/s41388-023-02841-0 |
| 45 |
doi: 10.1038/s41392-025-02302-y |
| 46 |
doi: 10.1186/s12943-024-02008-9 |
| 47 |
doi: 10.1016/j.canlet.2023.216285 |
| 48 |
doi: 10.1016/j.yexcr.2022.113439 |
| 49 |
doi: 10.15252/embr.202256052 |
| 50 |
doi: 10.1016/j.tranon.2023.101758 |
| 51 |
doi: 10.1038/s41392-021-00595-3 |
| 52 |
doi: 10.3390/cancers14092306 |
| 53 |
doi: 10.3892/or.2022.8288 |
| 54 |
doi: 10.1038/onc.2017.6 |
| 55 |
doi: 10.3389/fonc.2021.632364 |
| 56 |
doi: 10.1038/s41586-019-0977-x |
| 57 |
doi: 10.1038/s41416-020-0727-8 |
| 58 |
doi: 10.1016/j.ejmech.2020.112861 |
| 59 |
doi: 10.1038/s41408-018-0154-8 |
| 60 |
doi: 10.1111/bjh.12819 |
| 61 |
doi: 10.1200/JCO.2014.59.2782 |
| 62 |
doi: 10.3109/10428194.2015.1137292 |
| 63 |
doi: 10.1016/j.phrs.2023.106868 |
| 64 |
doi: 10.1126/sciadv.adg4993 |
| [1] | Hai WANG, Yizhou JIANG. Molecular mechanisms and clinical applications of anti-angiogenic therapy in precision treatment of breast cancer [J]. Journal of Peking University (Health Sciences), 2026, 58(2): 251-256. |
| [2] | Bixian LUO, Hongming LIU, Weixun XIE, Weihua GONG. Novel clinical insights and frontier issues in alpha- fetoprotein-producing gastric cancer [J]. Journal of Peking University (Health Sciences), 2026, 58(2): 257-265. |
| [3] | Wen DU, Wenbo ZHANG, Yao YU, Shuo LIU, Huiyu SU, Leihao HU, Zunan TANG, Binzhang WU, Zhen CHEN, Jiaqi LI, Hao WANG, Xin PENG. Exploration and clinical application of the "digital and intelligent surgery" diagnosis and treatment workflow for oral and maxillofacial tumors [J]. Journal of Peking University (Health Sciences), 2026, 58(2): 278-284. |
| [4] | Ruofan GAO, Tianyu MA, Runkai WANG, Yuchen YIN, Ruidi LI, Dandan WANG, Bin XIA. Anti-inflammatory effects of cell membrane vesicle-mediated delivery of small interfering RNA targeting tumor necrosis factor-α on dental pulp stem cells [J]. Journal of Peking University (Health Sciences), 2026, 58(1): 22-29. |
| [5] | Boda GUO, Min LU, Guoliang WANG, Hongxian ZHANG, Lei LIU, Xiaofei HOU, Lei ZHAO, Xiaojun TIAN, Shudong ZHANG. Clinicopathological and prognostic differences between clear cell and non-clear cell renal cell carcinoma with venous tumor thrombus [J]. Journal of Peking University (Health Sciences), 2025, 57(4): 644-649. |
| [6] | Zhanyi ZHANG, Min LU, Yuehao SUN, Jinghan DONG, Xiaofei HOU, Chunlei XIAO, Guoliang WANG, Xiaojun TIAN, Lulin MA, Hongxian ZHANG, Shudong ZHANG. Clinicopathological features and survival analysis of TFE3-rearranged renal cell carcinoma with venous tumor thrombus [J]. Journal of Peking University (Health Sciences), 2025, 57(4): 650-661. |
| [7] | Qi MIAO, Baoan HONG, Xuezhou ZHANG, Zhipeng SUN, Wei WANG, Yuxuan WANG, Yuxuan BO, Jiahui ZHAO, Ning ZHANG. Risk assessment of perioperative adverse events and management of antiplatelet therapy in patients with bladder cancer and coronary atherosclerotic heart disease undergoing transurethral resection of bladder cancer [J]. Journal of Peking University (Health Sciences), 2025, 57(4): 698-703. |
| [8] | Shuai LIU, Zhuo LIU, Yunhe GUAN, Guoliang WANG, Xiaojun TIAN, Hongxian ZHANG, Lei LIU, Lulin MA, Shudong ZHANG. Robot-assisted laparoscopic inferior vena cava segmental resection for renal tumor with tumor thrombus invading the vascular wall [J]. Journal of Peking University (Health Sciences), 2025, 57(4): 796-802. |
| [9] | Weihao LI, Jing LI, Xuemin ZHANG, Wei LI, Qingle LI, Xiaoming ZHANG. Effect of intraoperative blood salvage autotransfusion on the prognosis of patients after carotid body tumor resection [J]. Journal of Peking University (Health Sciences), 2025, 57(2): 272-276. |
| [10] | Yuanyuan YANG, Shanshan ZHANG, Guangyan YU, Huijun YANG, Hongyu YANG. Clinical outcomes of partial sialoadenectomy for the treatment of benign tumors in the submandibular gland [J]. Journal of Peking University (Health Sciences), 2025, 57(2): 334-339. |
| [11] | Suqing TIAN, Haitao SUN, Tiandi ZHAO, Wei WANG. Analysis of positioning errors in head and neck cancers during radiotherapy assisted by the 6D treatment couch and image-guided radiation therapy [J]. Journal of Peking University (Health Sciences), 2024, 56(6): 1097-1100. |
| [12] | Jiajun LIU, Guokang LIU, Yuhu ZHU. Immune-related severe pneumonia: A case report [J]. Journal of Peking University (Health Sciences), 2024, 56(5): 932-937. |
| [13] | Kewei CHEN,Zhuo LIU,Shaohui DENG,Fan ZHANG,Jianfei YE,Guoliang WANG,Shudong ZHANG. Clinical diagnosis and treatment of renal angiomyolipoma with inferior vena cava tumor thrombus [J]. Journal of Peking University (Health Sciences), 2024, 56(4): 617-623. |
| [14] | Shuai LIU,Lei LIU,Zhuo LIU,Fan ZHANG,Lulin MA,Xiaojun TIAN,Xiaofei HOU,Guoliang WANG,Lei ZHAO,Shudong ZHANG. Clinical treatment and prognosis of adrenocortical carcinoma with venous tumor thrombus [J]. Journal of Peking University (Health Sciences), 2024, 56(4): 624-630. |
| [15] | Jie YANG,Jieli FENG,Shudong ZHANG,Lulin MA,Qing ZHENG. Clinical effects of transesophageal echocardiography in different surgical methods for nephrectomy combined with Mayo Ⅲ-Ⅳ vena tumor thrombectomy [J]. Journal of Peking University (Health Sciences), 2024, 56(4): 631-635. |
|
||