北京大学学报(医学版) ›› 2026, Vol. 58 ›› Issue (2): 290-300. doi: 10.19723/j.issn.1671-167X.2026.02.011
结直肠癌根治术后肝转移风险多中心列线图预测模型的构建与验证
王楠楠1,2,*, 袁大晋1,*, 朱昱冰1, 丁磊1,*( )
)
- 1. 首都医科大学附属北京世纪坛医院胃肠外科,北京 100038
2. 江苏省肿瘤医院(南京医科大学附属肿瘤医院,江苏省癌症中心)结直肠外科,南京 210009
Development and validation of a multicenter nomogram predicting the risk of liver metastasis after curative resection of colorectal cancer
Nannan WANG1,2, Dajin YUAN1, Yubing ZHU1, Lei DING1,*( )
)
- 1. Department of Gastrointestinal Surgery, Beijing Shijitan Hospital, Capital Medical University, Beijing 100038, China
2. Department of Colorectal Surgery, The Affiliated Cancer Hospital of Nanjing Medical University & Jiangsu Cancer Hospital & Jiangsu Institute of Cancer Research, Nanjing 210009, China
摘要:
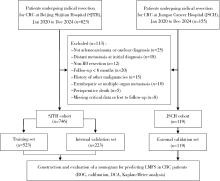
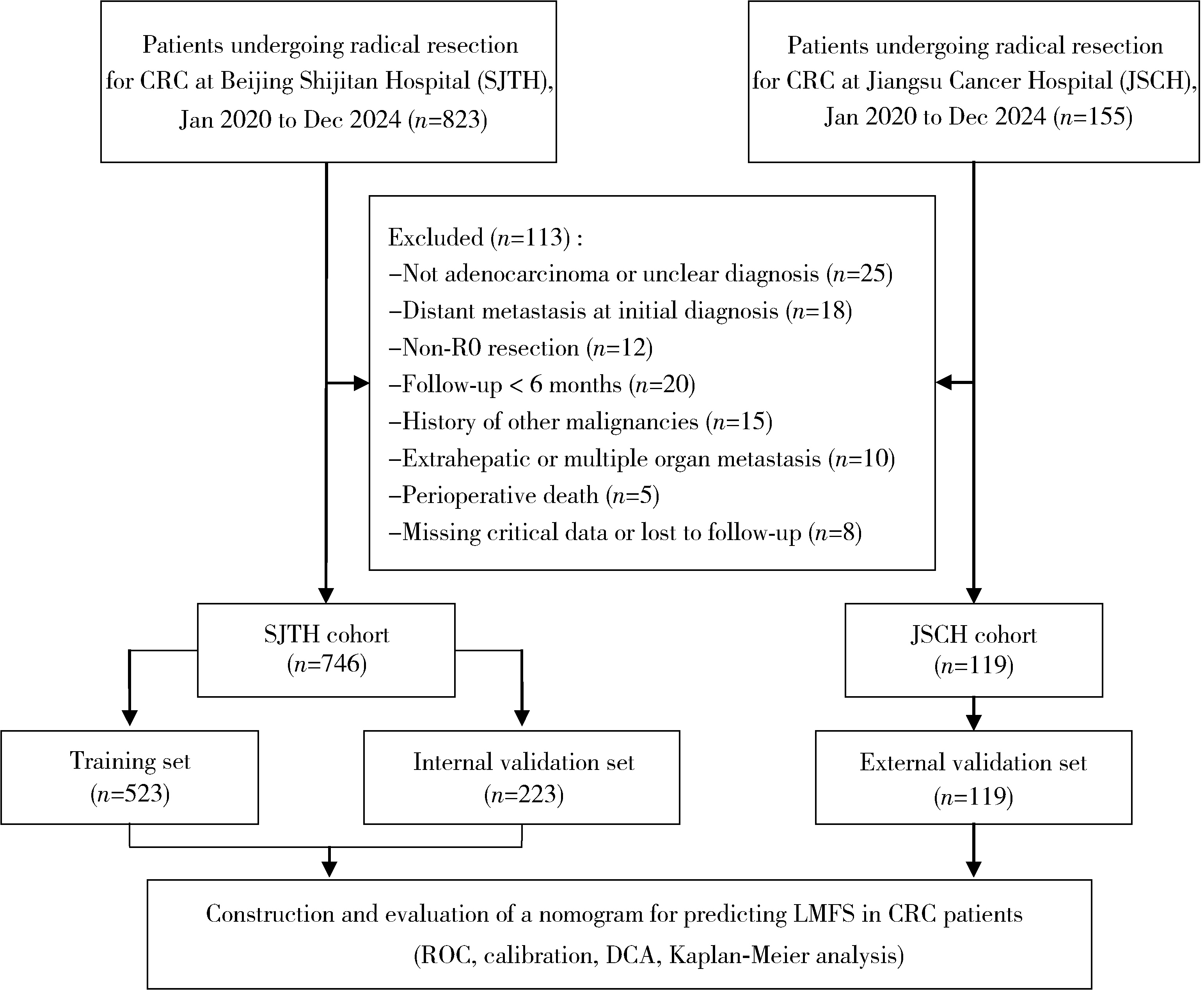
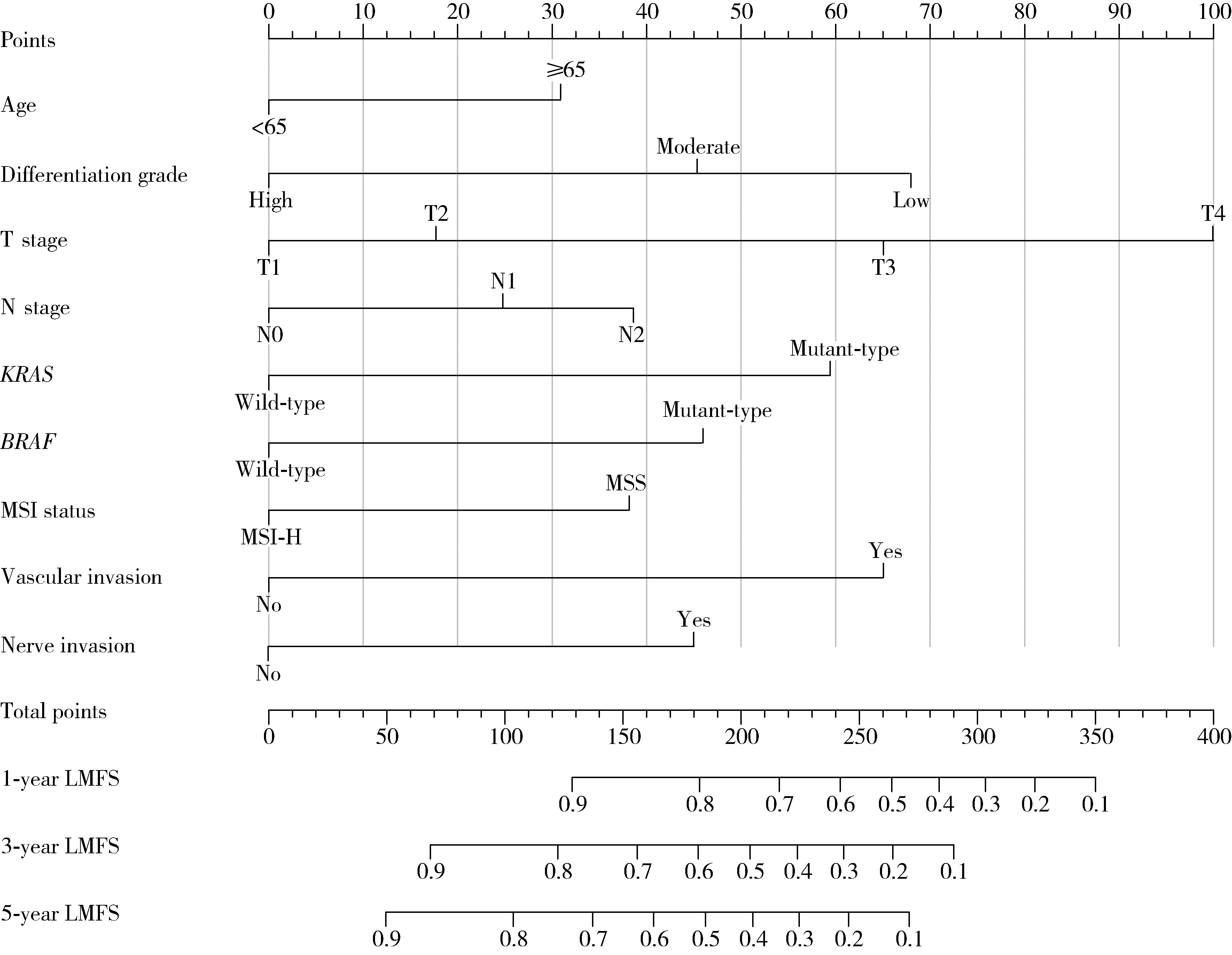
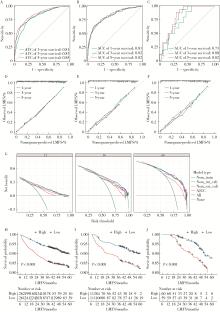
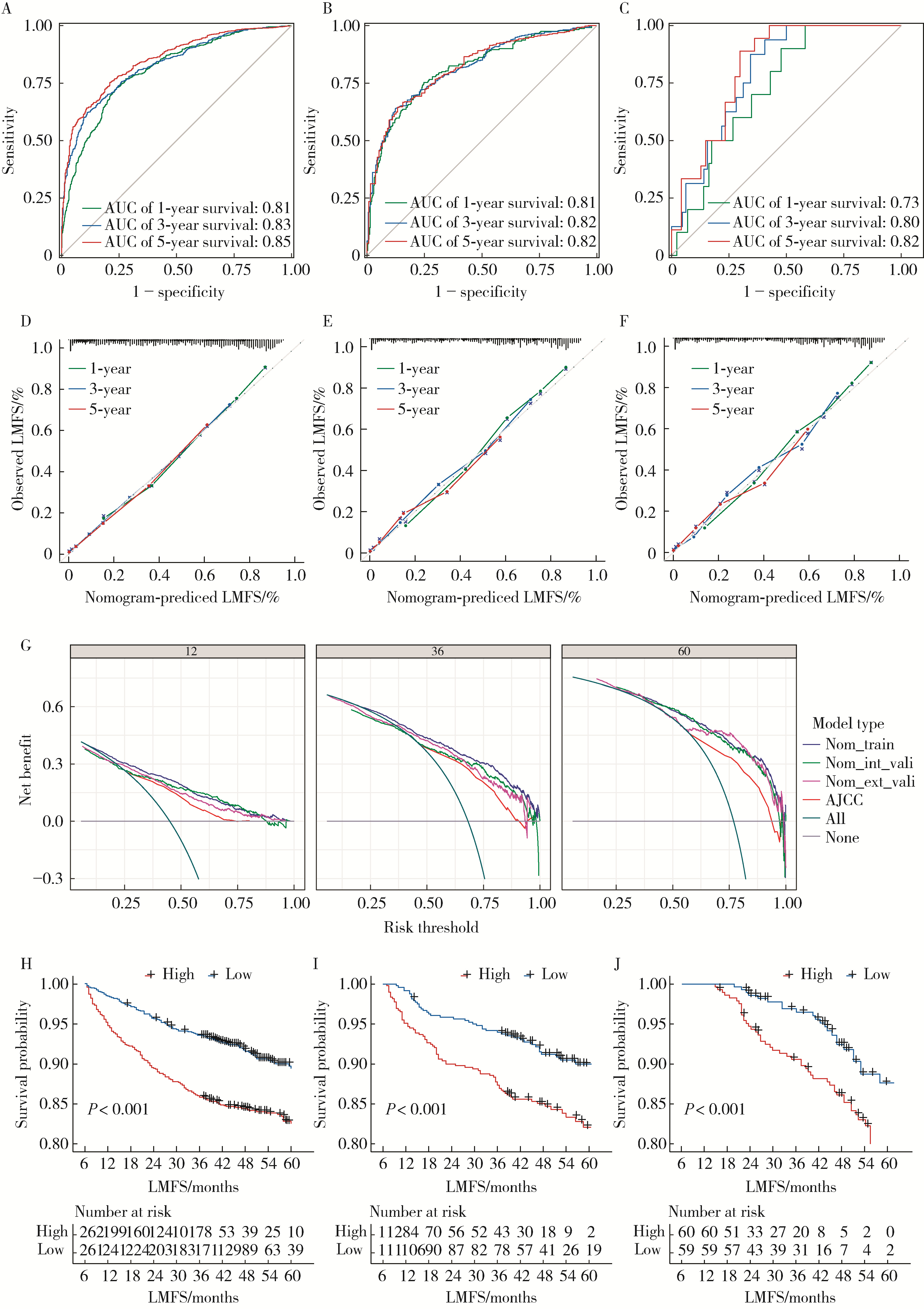
目的: 分析Ⅰ~Ⅲ期结直肠癌(colorectal cancer, CRC)根治性切除术后患者发生异时性肝转移的危险因素, 并构建用于预测患者术后1年、3年及5年无肝转移生存期(liver metastasis-free survival, LMFS)的列线图模型。方法: 采用多中心回顾性队列研究设计, 连续收集2020年1月至2024年12月于首都医科大学附属北京世纪坛医院行根治性切除术的746例CRC患者的病例资料, 按7 ∶3比例随机划分为训练集(523例)与内部验证集(223例); 连续选择同期江苏省肿瘤医院的119例患者作为独立外部验证集。纳入指标包括患者的临床病理特征及微卫星不稳定性(microsatellite instability, MSI)、KRAS/BRAF基因状态等分子标志物。采用单因素及多因素Cox比例风险回归分析筛选独立预测因子, 并据此构建LMFS列线图模型。通过一致性(concordance, C)指数、时间依赖性受试者工作特征(receiver operating characteristic, ROC)曲线的曲线下面积(area under the curve, AUC)、校准曲线及决策曲线分析(decision curve analysis, DCA)综合评估模型的区分度、校准度与临床实用性。结果: 研究的多中心队列基线资料均衡(P>0.05)。多因素分析显示, 高龄(≥65岁)、低分化、T分期进展、N分期进展、脉管侵犯、神经侵犯及分子标志物状态均为异时性肝转移的独立预后因素, 其中, KRAS突变(HR=1.42, 95%CI: 1.27~1.63)与BRAF突变(HR=1.53, 95%CI: 1.29~1.84)为异时性肝转移的独立危险因素, 而微卫星高度不稳定(microsatellite instability-high, MSI-H)状态(HR=0.71, 95%CI: 0.54~0.92)为异时性肝转移的独立保护因素。列线图模型在训练集、内部验证集和外部验证集中的C指数分别为0.85(95%CI: 0.82~0.89)、0.81(95%CI: 0.77~0.83)和0.75(95%CI: 0.71~0.79);训练集预测1年、3年、5年LMFS的AUC分别为0.81(95%CI: 0.77~0.86)、0.83(95%CI: 0.80~0.89)和0.85(95%CI: 0.78~0.92)。校准曲线显示预测值与实测值高度一致; DCA表明该预测模型相较于AJCC(American Joint Committee on Cancer)的TNM分期系统具有更高的临床净收益; 各队列中高风险组与低风险组的LMFS差异均具有统计学意义(P < 0.001)。结论: 本研究整合临床病理特征与KRAS、BRAF、MSI分子标志物构建的列线图模型, 在预测Ⅰ~Ⅲ期CRC患者术后异时性肝转移风险方面具有良好的区分度、校准度及临床实用性, 且优于传统TNM分期系统, 有助于指导术后个性化随访监测与治疗决策。
中图分类号:
- R735.34
| 1 |
|
| 2 |
|
| 3 |
doi: 10.1200/JCO.22.01690 |
| 4 |
doi: 10.1001/jamanetworkopen.2022.36666 |
| 5 |
doi: 10.1016/j.critrevonc.2023.103990 |
| 6 |
doi: 10.6004/jnccn.2021.0012 |
| 7 |
|
| 8 |
doi: 10.1016/j.surg.2022.10.014 |
| 9 |
doi: 10.1016/S1470-2045(14)71116-7 |
| 10 |
doi: 10.1186/s12967-019-1940-1 |
| 11 |
doi: 10.1097/JS9.0000000000000415 |
| 12 |
doi: 10.7150/jca.46155 |
| 13 |
|
| 14 |
doi: 10.1186/s12957-022-02558-6 |
| 15 |
doi: 10.3389/fonc.2022.844067 |
| 16 |
Shao S, Tian D, Li M, et al. Survival prediction in sigmoid-colon-cancer patients with liver metastasis: A prospective cohort study[J/OL]. JNCI Cancer Spectr, 2024, 8(5): pkae080[2025-11-01]. https://doi.org/10.1093/jncics/pkae080.
|
| 17 |
|
| 18 |
doi: 10.21037/jgo-2025-415 |
| 19 |
doi: 10.1038/s41467-018-04278-6 |
| 20 |
doi: 10.1186/s13045-024-01627-5 |
| 21 |
doi: 10.1001/jamasurg.2018.0996 |
| 22 |
doi: 10.1038/s41408-023-00833-7 |
| 23 |
doi: 10.1093/annonc/mdz139 |
| 24 |
doi: 10.1016/j.tranon.2024.101997 |
| 25 |
|
| 26 |
doi: 10.1016/j.ijsu.2017.04.068 |
| [1] | 李博闻, 张强, 孙益鑫. 儿童及青年漏斗胸患者Nuss术后发生脊柱侧弯的风险预测模型建立及验证[J]. 北京大学学报(医学版), 2025, 57(5): 941-946. |
| [2] | 韩丹, 次仁央金, 李秋红, 李军. 高海拔地区结直肠良恶性肿瘤患者肠道菌群差异及其与低海拔地区正常人群的比较[J]. 北京大学学报(医学版), 2025, 57(3): 578-583. |
| [3] | 刘园梅, 傅义程, 郝靖欣, 张福春, 刘慧琳. 老年髋部骨折患者住院期间发生术后心力衰竭的列线图预测模型的构建及验证[J]. 北京大学学报(医学版), 2024, 56(5): 874-883. |
| [4] | 李志存, 吴天俣, 梁磊, 范宇, 孟一森, 张骞. 穿刺活检单针阳性前列腺癌术后病理升级的危险因素分析及列线图模型构建[J]. 北京大学学报(医学版), 2024, 56(5): 896-901. |
| [5] | 于书慧,韩佳凝,钟丽君,陈聪语,肖云翔,黄燕波,杨洋,车新艳. 术前盆底肌电生理参数对前列腺癌根治性切除术后早期尿失禁的预测价值[J]. 北京大学学报(医学版), 2024, 56(4): 594-599. |
| [6] | 何海龙,李清,徐涛,张晓威. 构建显微精索手术治疗精索疼痛的术后疼痛缓解预测模型[J]. 北京大学学报(医学版), 2024, 56(4): 646-655. |
| [7] | 周泽臻,邓绍晖,颜野,张帆,郝一昌,葛力源,张洪宪,王国良,张树栋. 非转移性T3a肾细胞癌患者3年肿瘤特异性生存期预测[J]. 北京大学学报(医学版), 2024, 56(4): 673-679. |
| [8] | 苏俊琪,王晓颖,孙志强. 舌鳞状细胞癌根治性切除术后患者预后预测列线图的构建与验证[J]. 北京大学学报(医学版), 2024, 56(1): 120-130. |
| [9] | 毛海,张帆,张展奕,颜野,郝一昌,黄毅,马潞林,褚红玲,张树栋. 基于MRI前列腺腺体相关参数构建腹腔镜前列腺癌术后尿失禁的预测模型[J]. 北京大学学报(医学版), 2023, 55(5): 818-824. |
| [10] | 张云静,乔丽颖,祁萌,严颖,亢伟伟,刘国臻,王明远,席云峰,王胜锋. 乳腺癌患者新发心血管疾病预测模型的建立与验证:基于内蒙古区域医疗数据[J]. 北京大学学报(医学版), 2023, 55(3): 471-479. |
| [11] | 梁丽,李鑫,农琳,董颖,张继新,李东,李挺. 子宫内膜癌微卫星不稳定性分析: 微小微卫星变换的意义[J]. 北京大学学报(医学版), 2023, 55(2): 254-261. |
| [12] | 丁婷婷,曾楚雄,胡丽娜,余明华. 基于癌症基因组图谱数据库结直肠癌免疫细胞浸润预测模型的建立[J]. 北京大学学报(医学版), 2022, 54(2): 203-208. |
| [13] | 刘承,马潞林. 前入路机器人辅助前列腺根治性切除术中改善排尿控制的经验[J]. 北京大学学报(医学版), 2021, 53(4): 635-639. |
| [14] | 王文鹏,王捷夫,胡均,王俊锋,刘嘉,孔大陆,李健. 结直肠间质瘤临床病理特征及预后分析[J]. 北京大学学报(医学版), 2020, 52(2): 353-361. |
| [15] | 张旭初,张建华,王荣福,范岩,付占立,闫平,赵光宇,白艳霞. 18F-FDG PET/CT联合多种肿瘤标志物在结直肠中分化腺癌术后复发及转移中的应用价值[J]. 北京大学学报(医学版), 2019, 51(6): 1071-1077. |
|
||