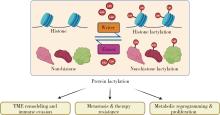
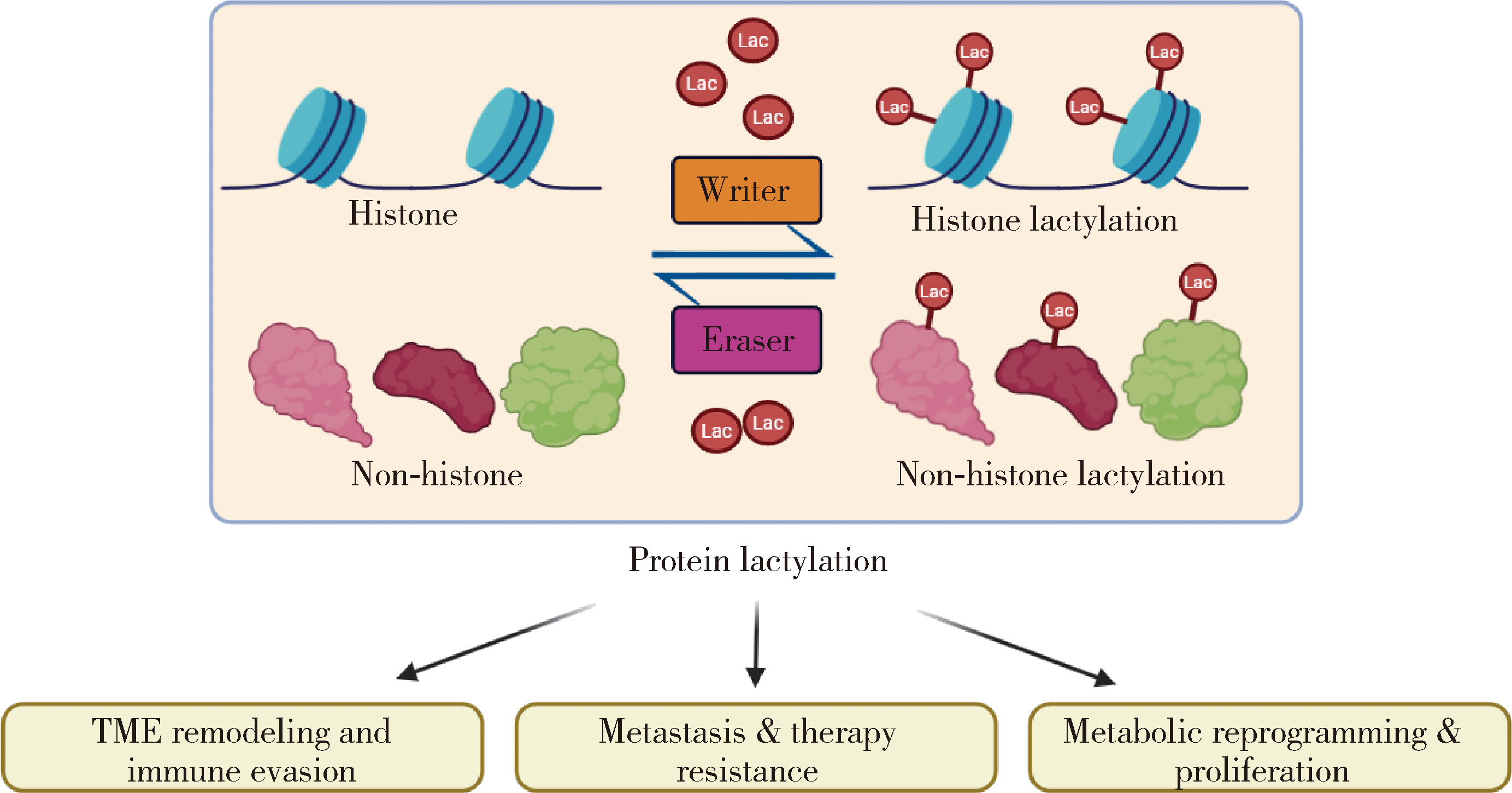
| 1 |
Brooks GA , Osmond AD , Arevalo JA , et al. Lactate as a myokine and exerkine: Drivers and signals of physiology and metabolism[J]. J Appl Physiol, 2023, 134 (3): 529- 548.
doi: 10.1152/japplphysiol.00497.2022
|
| 2 |
Certo M , Llibre A , Lee W , et al. Understanding lactate sensing and signalling[J]. Trends Endocrinol Metab, 2022, 33 (10): 722- 735.
doi: 10.1016/j.tem.2022.07.004
|
| 3 |
Li X , Yang Y , Zhang B , et al. Lactate metabolism in human health and disease[J]. Signal Transduct Target Ther, 2022, 7 (1): 305.
doi: 10.1038/s41392-022-01151-3
|
| 4 |
Brooks GA . Lactate as a fulcrum of metabolism[J]. Redox Biol, 2020, 35, 101454.
doi: 10.1016/j.redox.2020.101454
|
| 5 |
Hou X , Hong Z , Zeng H , et al. Lactylation in cancer biology: Unlocking new avenues for research and therapy[J]. Cancer Commun, 2025, 45 (11): 1367- 1406.
doi: 10.1002/cac2.70054
|
| 6 |
Wishart DS . Is cancer a genetic disease or a metabolic disease?[J]. EBioMedicine, 2015, 2 (6): 478- 479.
doi: 10.1016/j.ebiom.2015.05.022
|
| 7 |
Racker E . Bioenergetics and the Problem of Tumor Growth: An understanding of the mechanism of the generation and control of biological energy may shed light on the problem of tumor growth[J]. Am Sci, 1972, 60 (1): 56- 63.
|
| 8 |
de la Cruz-López KG , Castro-Muñoz LJ , Reyes-Hernández DO , et al. Lactate in the regulation of tumor microenvironment and therapeutic approaches[J]. Front Oncol, 2019, 9, 1143.
doi: 10.3389/fonc.2019.01143
|
| 9 |
Zhang D , Tang Z , Huang H , et al. Metabolic regulation of gene expression by histone lactylation[J]. Nature, 2019, 574 (7779): 575- 580.
doi: 10.1038/s41586-019-1678-1
|
| 10 |
Sun L , Zhang H , Gao P . Metabolic reprogramming and epigenetic modifications on the path to cancer[J]. Protein Cell, 2022, 13 (12): 877- 919.
doi: 10.1007/s13238-021-00846-7
|
| 11 |
Yao W , Hu X , Wang X . Crossing epigenetic frontiers: The intersection of novel histone modifications and diseases[J]. Signal Transduct Target Ther, 2024, 9 (1): 232.
doi: 10.1038/s41392-024-01918-w
|
| 12 |
Li Y , Zhang R , Hei H . Advances in post-translational modifications of proteins and cancer immunotherapy[J]. Front Immunol, 2023, 14, 1229397.
doi: 10.3389/fimmu.2023.1229397
|
| 13 |
Xu H , Wu M , Ma X , et al. Function and mechanism of novel histone posttranslational modifications in health and disease[J]. Biomed Res Int, 2021, 2021, 6635225.
doi: 10.1155/2021/6635225
|
| 14 |
Fu Y , Yu J , Li F , et al. Oncometabolites drive tumorigenesis by enhancing protein acylation: From chromosomal remodelling to nonhistone modification[J]. J Exp Clin Cancer Res, 2022, 41 (1): 144.
doi: 10.1186/s13046-022-02338-w
|
| 15 |
Wan N , Wang N , Yu S , et al. Cyclic immonium ion of lactyl-lysine reveals widespread lactylation in the human proteome[J]. Nat Meth, 2022, 19 (7): 854- 864.
doi: 10.1038/s41592-022-01523-1
|
| 16 |
Yang YH , Wang QC , Kong J , et al. Global profiling of lysine lactylation in human lungs[J]. Proteomics, 2023, 23 (15): e2200437.
doi: 10.1002/pmic.202200437
|
| 17 |
Wu X . In-depth discovery of protein lactylation in hepatocellular carcinoma[J]. Proteomics, 2023, 23 (9): e2300003.
doi: 10.1002/pmic.202300003
|
| 18 |
Hong H , Chen X , Wang H , et al. Global profiling of protein lysine lactylation and potential target modified protein analysis in hepatocellular carcinoma[J]. Proteomics, 2023, 23 (9): e2200432.
doi: 10.1002/pmic.202200432
|
| 19 |
Lin J , Liu G , Chen L , et al. Targeting lactate-related cell cycle activities for cancer therapy[J]. Semin Cancer Biol, 2022, 86 (Pt 3): 1231- 1243.
|
| 20 |
Yang D , Yin J , Shan L , et al. Identification of lysine-lactylated substrates in gastric cancer cells[J]. iScience, 2022, 25 (7): 104630.
doi: 10.1016/j.isci.2022.104630
|
| 21 |
Jiang P , Ning W , Shi Y , et al. FSL-Kla: A few-shot learning-based multi-feature hybrid system for lactylation site prediction[J]. Comput Struct Biotechnol J, 2021, 19, 4497- 4509.
doi: 10.1016/j.csbj.2021.08.013
|
| 22 |
Liberti MV , Locasale JW . Histone lactylation: A new role for glucose metabolism[J]. Trends Biochem Sci, 2020, 45 (3): 179- 182.
doi: 10.1016/j.tibs.2019.12.004
|
| 23 |
Wang P , Xie D , Xiao T , et al. H3K18 lactylation promotes the progression of arsenite-related idiopathic pulmonary fibrosis via YTHDF1/m6A/NREP[J]. J Hazard Mater, 2024, 461, 132582.
doi: 10.1016/j.jhazmat.2023.132582
|
| 24 |
Li L , Chen K , Wang T , et al. Glis1 facilitates induction of pluripotency via an epigenome-metabolome-epigenome signalling cascade[J]. Nat Metab, 2020, 2 (9): 882- 892.
doi: 10.1038/s42255-020-0267-9
|
| 25 |
Kozlakidis Z , Shi P , Abarbanel G , et al. Recent developments in protein lactylation in PTSD and CVD: Novel strategies and targets[J]. BioTech, 2023, 12 (2): 38.
doi: 10.3390/biotech12020038
|
| 26 |
Li R , Yang Y , Wang H , et al. Lactate and lactylation in the brain: Current progress and perspectives[J]. Cell Mol Neurobiol, 2023, 43 (6): 2541- 2555.
doi: 10.1007/s10571-023-01335-7
|
| 27 |
Zong Z , Xie F , Wang S , et al. Alanyl-tRNA synthetase, AARS1, is a lactate sensor and lactyltransferase that lactylates p53 and contributes to tumorigenesis[J]. Cell, 2024, 187 (10): 2375- 2392.e33.
doi: 10.1016/j.cell.2024.04.002
|
| 28 |
Chen H , Li Y , Li H , et al. NBS1 lactylation is required for efficient DNA repair and chemotherapy resistance[J]. Nature, 2024, 631 (8021): 663- 669.
doi: 10.1038/s41586-024-07620-9
|
| 29 |
Chen Y , Wu J , Zhai L , et al. Metabolic regulation of homologous recombination repair by MRE11 lactylation[J]. Cell, 2024, 187 (2): 294- 311.
doi: 10.1016/j.cell.2023.11.022
|
| 30 |
Ali I , Conrad RJ , Verdin E , et al. Lysine acetylation goes global: From epigenetics to metabolism and therapeutics[J]. Chem Rev, 2018, 118 (3): 1216- 1252.
doi: 10.1021/acs.chemrev.7b00181
|
| 31 |
Moreno-Yruela C , Zhang D , Wei W , et al. Class Ⅰ histone deacetylases (HDAC1-3) are histone lysine delactylases[J]. Sci Adv, 2022, 8 (3): eabi6696.
doi: 10.1126/sciadv.abi6696
|
| 32 |
Walenta S , Wetterling M , Lehrke M , et al. High lactate levels predict likelihood of metastases, tumor recurrence, and restricted patient survival in human cervical cancers[J]. Cancer Res, 2000, 60 (4): 916- 921.
|
| 33 |
Ewaschuk JB , Naylor JM , Zello GA . D-lactate in human and ruminant metabolism[J]. J Nutr, 2005, 135 (7): 1619- 1625.
doi: 10.1093/jn/135.7.1619
|
| 34 |
Varner EL , Trefely S , Bartee D , et al. Quantification of lactoyl-CoA (lactyl-CoA) by liquid chromatography mass spectrometry in mammalian cells and tissues[J]. Open Biol, 2020, 10 (9): 200187.
doi: 10.1098/rsob.200187
|
| 35 |
Gaffney DO , Jennings EQ , Anderson CC , et al. Non-enzymatic lysine lactoylation of glycolytic enzymes[J]. Cell Chem Biol, 2020, 27 (2): 206- 213.
doi: 10.1016/j.chembiol.2019.11.005
|
| 36 |
Yang T , Zhang S , Nie K , et al. ZNF207-driven PRDX1 lactylation and NRF2 activation in regorafenib resistance and ferroptosis evasion[J]. Drug Resist Updat, 2025, 82, 101274.
doi: 10.1016/j.drup.2025.101274
|
| 37 |
Liu R , Wu J , Guo H , et al. Post-translational modifications of histones: Mechanisms, biological functions, and therapeutic targets[J]. MedComm, 2023, 4 (3): e292.
doi: 10.1002/mco2.292
|
| 38 |
Huang ZW , Zhang XN , Zhang L , et al. STAT5 promotes PD-L1 expression by facilitating histone lactylation to drive immuno-suppression in acute myeloid leukemia[J]. Signal Transduct Target Ther, 2023, 8 (1): 391.
doi: 10.1038/s41392-023-01605-2
|
| 39 |
De Leo A , Ugolini A , Yu X , et al. Glucose-driven histone lactylation promotes the immunosuppressive activity of monocyte-derived macrophages in glioblastoma[J]. Immunity, 2024, 57 (5): 1105- 1123.
doi: 10.1016/j.immuni.2024.04.006
|
| 40 |
Sun T , Liu B , Li Y , et al. Oxamate enhances the efficacy of CAR-T therapy against glioblastoma via suppressing ectonucleotidases and CCR8 lactylation[J]. J Exp Clin Cancer Res, 2023, 42 (1): 253.
doi: 10.1186/s13046-023-02815-w
|
| 41 |
Gu J , Zhou J , Chen Q , et al. Tumor metabolite lactate promotes tumorigenesis by modulating MOESIN lactylation and enhancing TGF-β signaling in regulatory T cells[J]. Cell Rep, 2022, 40 (3): 111122.
doi: 10.1016/j.celrep.2022.111122
|
| 42 |
Yang J , Yu X , Xiao M , et al. Histone lactylation-driven feedback loop modulates cholesterol-linked immunosuppression in pancreatic cancer[J]. Gut, 2025, 74 (11): e334361.
|
| 43 |
Wang J , Liu Z , Xu Y , et al. Enterobacterial LPS-inducible LINC00152 is regulated by histone lactylation and promotes cancer cells invasion and migration[J]. Front Cell Infect Microbiol, 2022, 12, 913815.
doi: 10.3389/fcimb.2022.913815
|
| 44 |
Zhou J , Xu W , Wu Y , et al. GPR37 promotes colorectal cancer liver metastases by enhancing the glycolysis and histone lactylation via Hippo pathway[J]. Oncogene, 2023, 42 (45): 3319- 3330.
doi: 10.1038/s41388-023-02841-0
|
| 45 |
Li X , Zhang C , Mei Y , et al. Irinotecan alleviates chemoresistance to anthracyclines through the inhibition of AARS1-mediated BLM lactylation and homologous recombination repair[J]. Signal Transduct Target Ther, 2025, 10 (1): 214.
doi: 10.1038/s41392-025-02302-y
|
| 46 |
Li F , Si W , Xia L , et al. Positive feedback regulation between glycolysis and histone lactylation drives oncogenesis in pancreatic ductal adenocarcinoma[J]. Mol Cancer, 2024, 23 (1): 90.
doi: 10.1186/s12943-024-02008-9
|
| 47 |
Chen M , Cen K , Song Y , et al. NUSAP1-LDHA-Glycolysis-Lactate feedforward loop promotes Warburg effect and metastasis in pancreatic ductal adenocarcinoma[J]. Cancer Lett, 2023, 567, 216285.
doi: 10.1016/j.canlet.2023.216285
|
| 48 |
Miao Z , Zhao X , Liu X . Hypoxia induced β-catenin lactylation promotes the cell proliferation and stemness of colorectal cancer through the Wnt signaling pathway[J]. Exp Cell Res, 2023, 422 (1): 113439.
doi: 10.1016/j.yexcr.2022.113439
|
| 49 |
Jin J , Bai L , Wang D , et al. SIRT3-dependent delactylation of cyclin E2 prevents hepatocellular carcinoma growth[J]. EMBO Rep, 2023, 24 (5): e56052.
doi: 10.15252/embr.202256052
|
| 50 |
Pandkar MR , Sinha S , Samaiya A , et al. Oncometabolite lactate enhances breast cancer progression by orchestrating histone lactylation-dependent c-Myc expression[J]. Transl Oncol, 2023, 37, 101758.
doi: 10.1016/j.tranon.2023.101758
|
| 51 |
Wu H , Wang Y , Ying M , et al. Lactate dehydrogenases amplify reactive oxygen species in cancer cells in response to oxidative stimuli[J]. Signal Transduct Target Ther, 2021, 6 (1): 242.
doi: 10.1038/s41392-021-00595-3
|
| 52 |
Maeda M , Ko M , Mane MM , et al. Genetic and drug inhibition of LDH-A: Effects on murine gliomas[J]. Cancers, 2022, 14 (9): 2306.
doi: 10.3390/cancers14092306
|
| 53 |
Zhang W , Wang C , Hu X , et al. Inhibition of LDHA suppresses cell proliferation and increases mitochondrial apoptosis via the JNK signaling pathway in cervical cancer cells[J]. Oncol Rep, 2022, 47 (4): 77.
doi: 10.3892/or.2022.8288
|
| 54 |
Jin L , Chun J , Pan C , et al. Phosphorylation-mediated activation of LDHA promotes cancer cell invasion and tumour metastasis[J]. Oncogene, 2017, 36 (27): 3797- 3806.
doi: 10.1038/onc.2017.6
|
| 55 |
Qiao T , Xiong Y , Feng Y , et al. Inhibition of LDHA by oxamate enhances the efficacy of anti-PD-1 treatment in an NSCLC humanized mouse model[J]. Front Oncol, 2021, 11, 632364.
doi: 10.3389/fonc.2021.632364
|
| 56 |
Elia I , Rossi M , Stegen S , et al. Breast cancer cells rely on environmental pyruvate to shape the metastatic niche[J]. Nature, 2019, 568 (7750): 117- 121.
doi: 10.1038/s41586-019-0977-x
|
| 57 |
McNeillis R , Greystoke A , Walton J , et al. A case of malignant hyperlactaemic acidosis appearing upon treatment with the mono-carboxylase transporter 1 inhibitor AZD3965[J]. Br J Cancer, 2020, 122 (8): 1141- 1145.
doi: 10.1038/s41416-020-0727-8
|
| 58 |
He ZX , Wei BF , Zhang X , et al. Current development of CBP/p300 inhibitors in the last decade[J]. Eur J Med Chem, 2021, 209, 112861.
doi: 10.1016/j.ejmech.2020.112861
|
| 59 |
Kaufman JL , Mina R , Jakubowiak AJ , et al. Combining carfilzomib and panobinostat to treat relapsed/refractory multiple myeloma: Results of a Multiple Myeloma Research Consortium Phase Ⅰ Study[J]. Blood Cancer J, 2019, 9 (1): 3.
doi: 10.1038/s41408-018-0154-8
|
| 60 |
Ogura M , Ando K , Suzuki T , et al. A multicentre phase Ⅱ study of vorinostat in patients with relapsed or refractory indolent B-cell non-Hodgkin lymphoma and mantle cell lymphoma[J]. Br J Haematol, 2014, 165 (6): 768- 776.
doi: 10.1111/bjh.12819
|
| 61 |
O'Connor OA , Horwitz S , Masszi T , et al. Belinostat in patients with relapsed or refractory peripheral T-cell lymphoma: Results of the pivotal phase Ⅱ BELIEF (CLN-19) study[J]. J Clin Oncol, 2015, 33 (23): 2492- 2499.
doi: 10.1200/JCO.2014.59.2782
|
| 62 |
Zinzani PL , Pellegrini C , Cerciello G , et al. Romidepsin in relapsed/refractory T-cell lymphomas: Italian experience and results of a named patient program[J]. Leuk Lymphoma, 2016, 57 (10): 2370- 2374.
doi: 10.3109/10428194.2015.1137292
|
| 63 |
Wang C , Feng F , Pan L , et al. Reply to the letter titled: Demethylzeylasteral targets lactate to suppress the tumorigenicity of liver cancer stem cells: Is it attributed to histone lactylation?[J]. Pharmacol Res, 2023, 194, 106868.
doi: 10.1016/j.phrs.2023.106868
|
| 64 |
Jia M , Yue X , Sun W , et al. ULK1-mediated metabolic reprogramming regulates Vps34 lipid kinase activity by its lactylation[J]. Sci Adv, 2023, 9 (22): eadg4993.
doi: 10.1126/sciadv.adg4993
|
 )
)