北京大学学报(医学版) ›› 2026, Vol. 58 ›› Issue (2): 257-265. doi: 10.19723/j.issn.1671-167X.2026.02.006
产甲胎蛋白胃癌的新临床特征和前沿科学问题
- 浙江大学医学院附属第二医院胃肠外科,杭州 310058
Novel clinical insights and frontier issues in alpha- fetoprotein-producing gastric cancer
Bixian LUO, Hongming LIU, Weixun XIE, Weihua GONG*( )
)
- Department of Gastrointestinal Surgery, Second Affiliated Hospital of Zhejiang University School of Medicine, Hangzhou 310058, China
摘要:
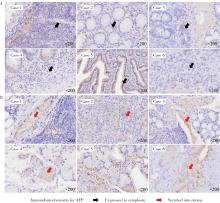
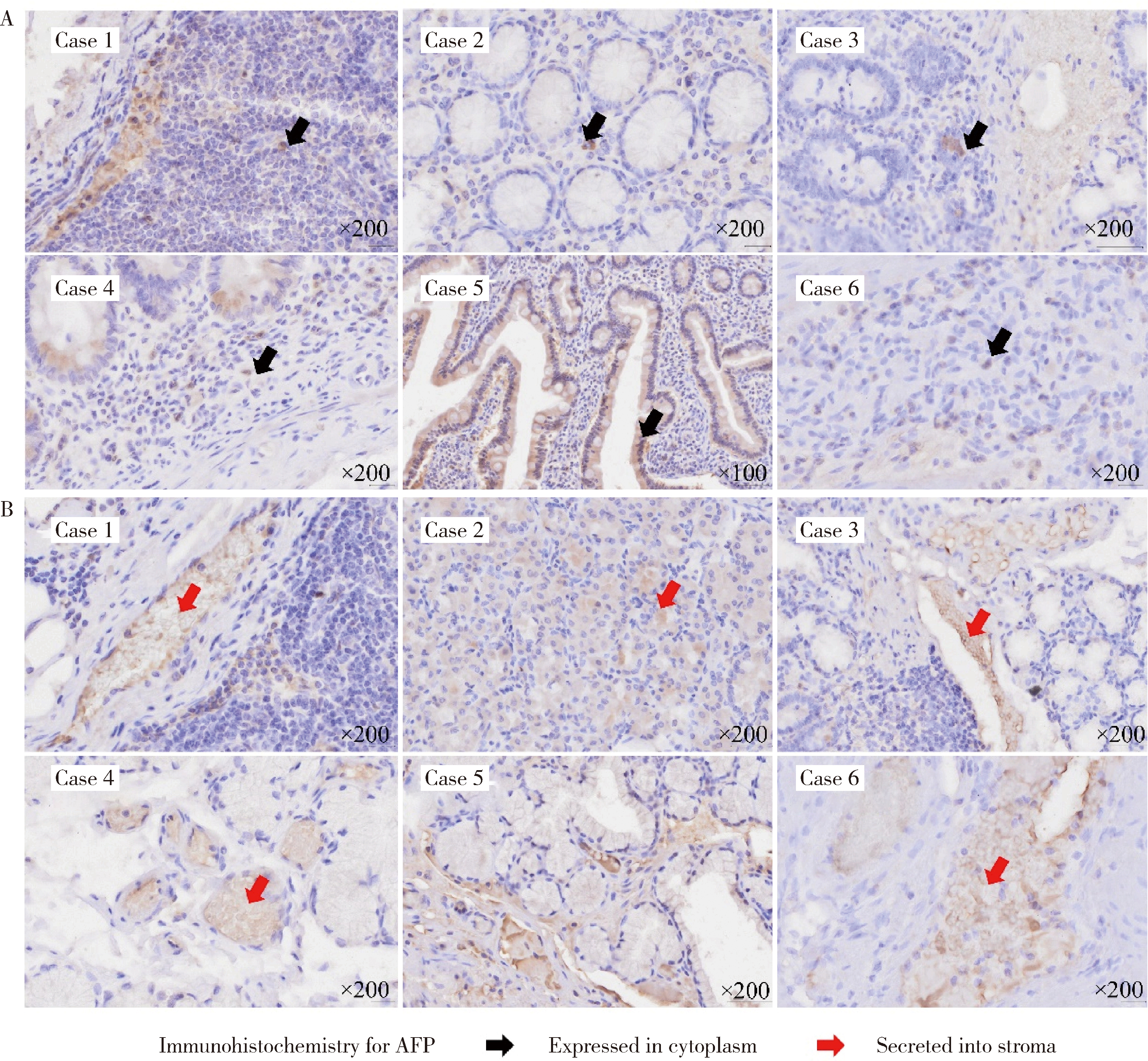
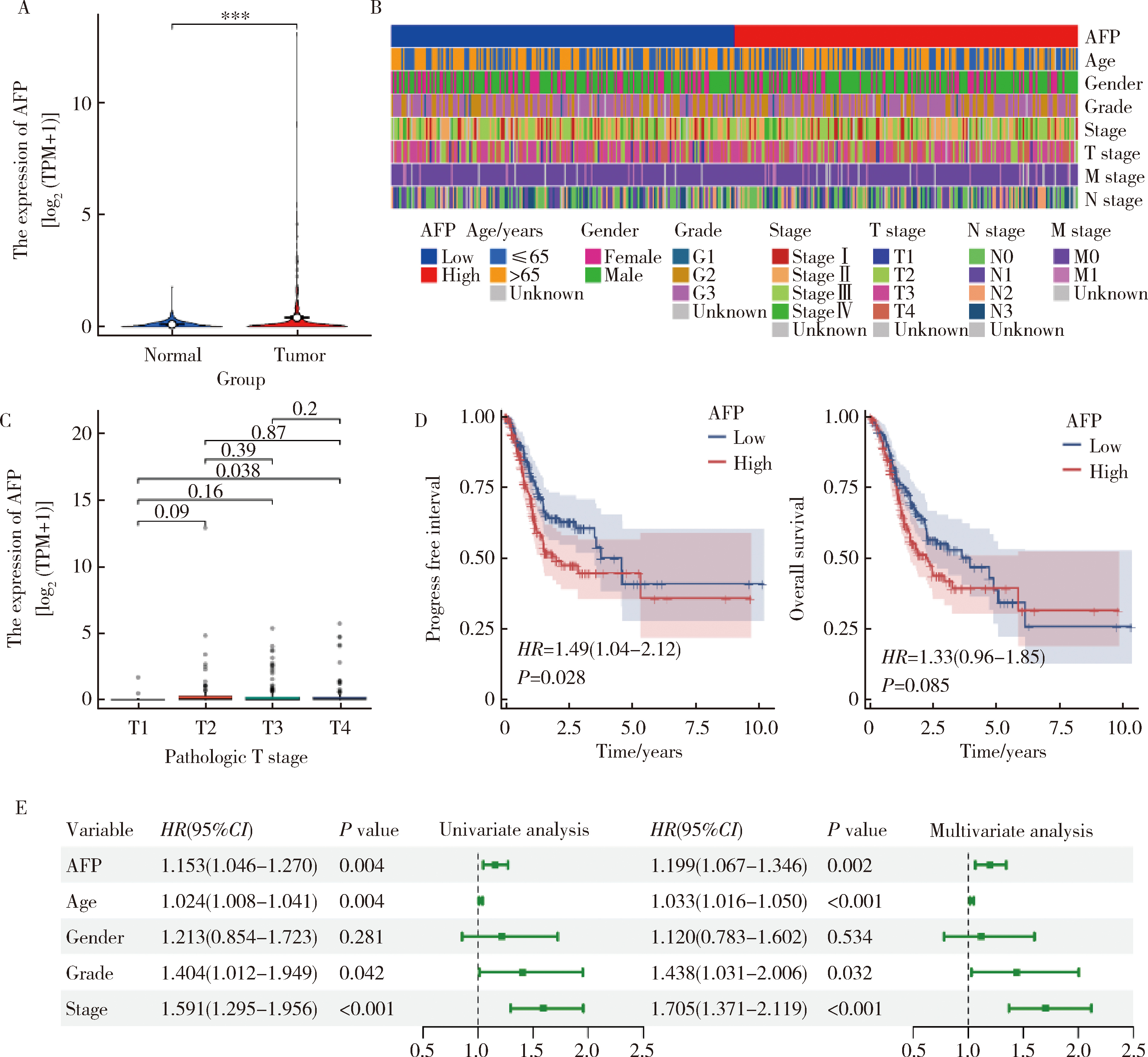
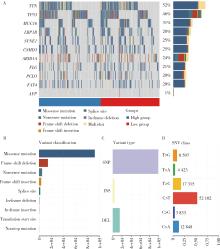
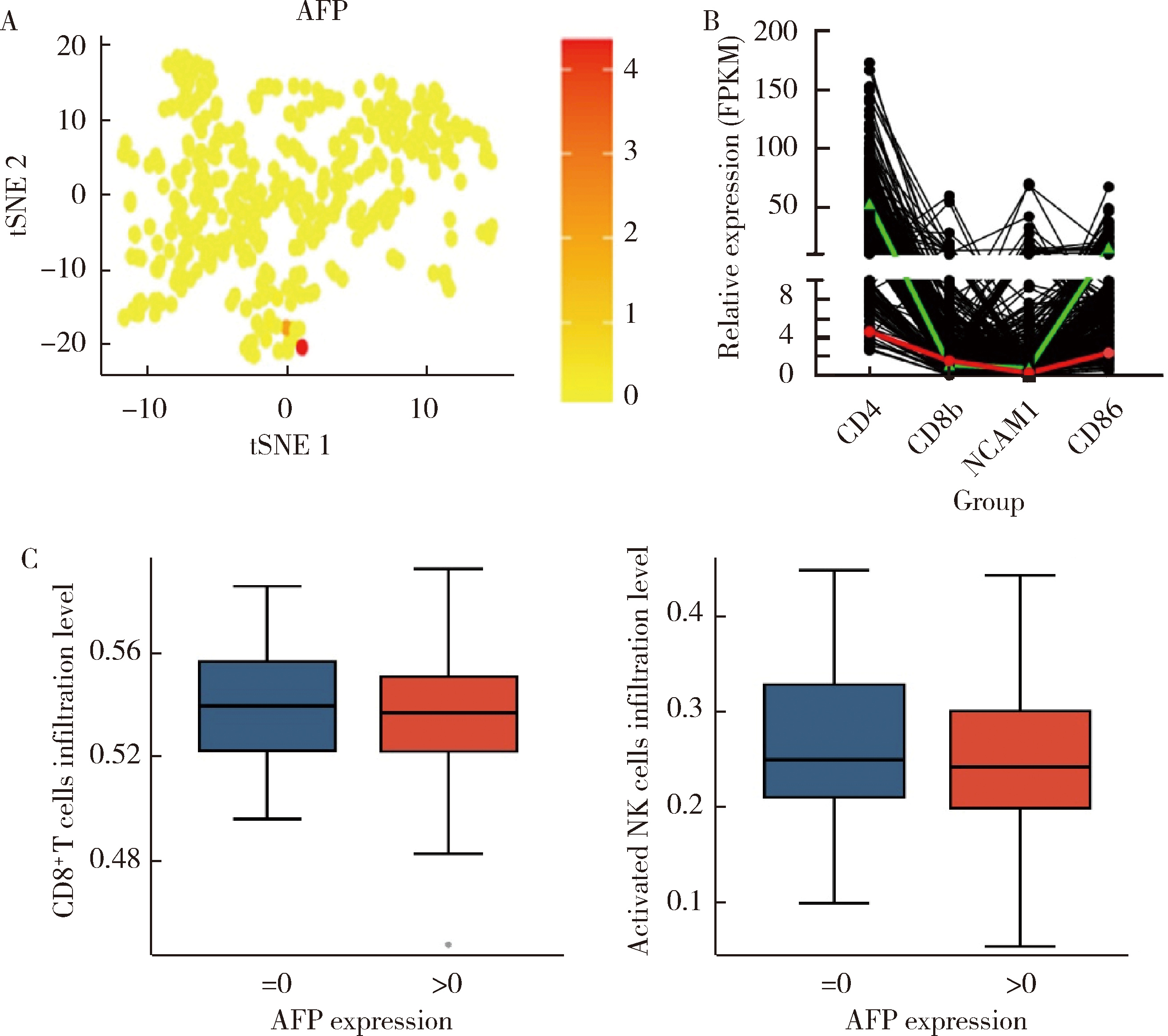
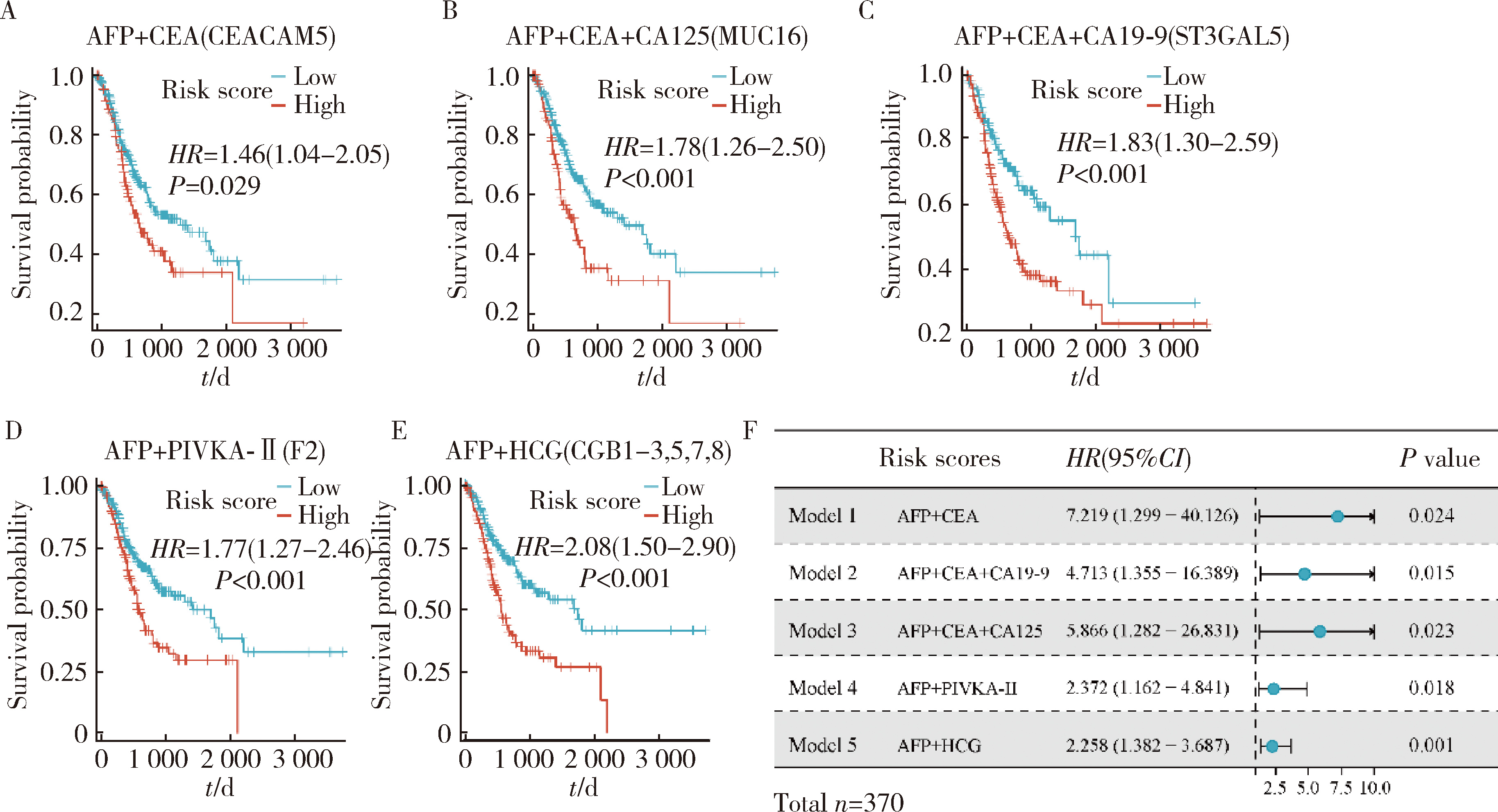
产甲胎蛋白胃癌(alpha-fetoprotein-producing gastric cancer,AFPGC)是一种特殊类型的胃癌,具有独特的生物学特性和临床表现。AFPGC大多数属于染色体不稳定型(chromosomal instability, CIN),其基因突变特征包括TP53、MUC16突变和ERBB2、周期蛋白CCNE1等的扩增。甲胎蛋白(alpha-fetoprotein, AFP)在胃癌组织中高表达,且与肿瘤T分期和患者预后相关。部分AFPGC患者虽血清AFP水平正常,但胃癌组织中AFP表达呈阳性。组织中的AFP在肿瘤的侵袭性和免疫逃逸中起着关键作用,通过调节免疫细胞功能,可能促使肿瘤生长和转移。临床上,AFP、癌胚抗原(carcinoembryonic antigen,CEA)、人绒毛膜促性腺素(human chorionic gonadotropin,HCG)、维生素K缺乏或拮抗剂-Ⅱ诱导的蛋白质(protein induced by vitamin K absence or antagonist-Ⅱ,PIVKA-Ⅱ,又称异常凝血酶原)等标志物的联合升高提示胃癌的高恶性潜能,尤其在晚期患者中,此类患者预后较差。免疫治疗在AFPGC中的应用显示出一定潜力,但疗效仍需进一步研究验证。尽管已有个案报告,但目前尚无统一的治疗方案,个体化、多模式治疗是提高AFPGC治疗效果的关键,因此,综合评估AFP水平、影像学和病理特征对临床治疗至关重要。
中图分类号:
- R735.2
| 1 |
doi: 10.1038/nature13480 |
| 2 |
doi: 10.1007/s12672-023-00731-1 |
| 3 |
doi: 10.3389/fonc.2022.962126 |
| 4 |
doi: 10.1016/j.canlet.2014.11.018 |
| 5 |
doi: 10.1038/s41388-021-01976-2 |
| 6 |
|
| 7 |
doi: 10.1186/s12876-023-02773-9 |
| 8 |
doi: 10.1111/apm.13196 |
| 9 |
doi: 10.3389/fonc.2021.633916 |
| 10 |
doi: 10.1007/s10120-019-00965-5 |
| 11 |
doi: 10.3390/cancers14163849 |
| 12 |
doi: 10.1007/s10120-025-01594-x |
| 13 |
王洁, 申璐璐, 张鑫, 等. 胃肝样腺癌临床病理及分子学特征分析[J]. 中华病理学杂志, 2025, 54 (7): 748- 754.
|
| 14 |
doi: 10.1002/cjp2.343 |
| 15 |
|
| 16 |
doi: 10.1186/s13578-025-01424-8 |
| 17 |
|
| 18 |
doi: 10.1073/pnas.80.15.4604 |
| 19 |
doi: 10.1038/s41467-021-24170-0 |
| 20 |
doi: 10.1136/jclinpath-2017-204928 |
| 21 |
doi: 10.1016/j.it.2022.04.001 |
| 22 |
doi: 10.3389/fonc.2024.1514069 |
| 23 |
|
| 24 |
|
| 25 |
|
| 26 |
|
| 27 |
doi: 10.2169/internalmedicine.31.260 |
| 28 |
doi: 10.3748/wjg.15.5106 |
| 29 |
doi: 10.47102/annals-acadmedsg.V30N1p58 |
| 30 |
|
| 31 |
doi: 10.2169/internalmedicine.43.926 |
| 32 |
|
| 33 |
|
| 34 |
|
| 35 |
|
| 36 |
doi: 10.3747/co.21.1768 |
| 37 |
doi: 10.3748/wjg.v24.i2.266 |
| 38 |
|
| 39 |
doi: 10.18632/oncotarget.22109 |
| 40 |
doi: 10.1186/s40064-016-3719-7 |
| 41 |
|
| 42 |
doi: 10.1007/s10147-022-02263-0 |
| 43 |
doi: 10.3389/fsurg.2023.1133335 |
| 44 |
doi: 10.1186/s12916-025-04047-5 |
| 45 |
doi: 10.1002/bjs.7081 |
| 46 |
doi: 10.1159/000065838 |
| [1] | 董琪, 何菁, 贾园, 姚海红, 张霞. 模拟复发性多软骨炎的VEXAS综合征1例[J]. 北京大学学报(医学版), 2025, 57(6): 1180-1183. |
| [2] | 张真伟, 徐欣然, 高学军, 董艳梅, 田华. RELT基因移码突变导致遗传性釉质发育不全[J]. 北京大学学报(医学版), 2025, 57(1): 13-18. |
| [3] | 金银姬, 刘蕊. 以肠系膜静脉血栓为突出表现的遗传性蛋白S缺乏症1例[J]. 北京大学学报(医学版), 2024, 56(6): 1106-1109. |
| [4] | 时云飞,王豪杰,刘卫平,米岚,龙孟平,刘雁飞,赖玉梅,周立新,刁新婷,李向红. 血管免疫母细胞性T细胞淋巴瘤临床与分子病理学特征分析[J]. 北京大学学报(医学版), 2023, 55(3): 521-529. |
| [5] | 熊焰,张波,聂立功,吴世凯,赵虎,李东,邸吉廷. 胸部SMARCA4缺失性未分化肿瘤的病理诊断与联合免疫检测点抑制剂治疗[J]. 北京大学学报(医学版), 2023, 55(2): 351-356. |
| [6] | 周秋君,龚潘,焦莶如,杨志仙. 1例Angelman综合征合并眼皮肤白化病2型患者的临床和遗传学分析及文献回顾[J]. 北京大学学报(医学版), 2023, 55(1): 181-185. |
| [7] | 程晓静,蒋栋,张连海,王江华,李雅真,翟佳慧,闫宝琪,张露露,谢兴旺,李子禹,季加孚. KRAS G12V特异性T细胞受体治疗恶性肿瘤的临床前研究[J]. 北京大学学报(医学版), 2022, 54(5): 884-895. |
| [8] | 秦彩朋,宋宇轩,丁梦婷,王飞,林佳兴,杨文博,杜依青,李清,刘士军,徐涛. 肾癌免疫治疗疗效评估突变预测模型的建立[J]. 北京大学学报(医学版), 2022, 54(4): 663-668. |
| [9] | 陈曦,王斯悦,薛恩慈,王雪珩,彭和香,范梦,王梦莹,武轶群,秦雪英,李劲,吴涛,朱洪平,李静,周治波,陈大方,胡永华. 基于核心家系全外显子组测序数据探索新生突变与非综合征型唇腭裂的关联[J]. 北京大学学报(医学版), 2022, 54(3): 387-393. |
| [10] | 曹泽,王乐童,刘振明. 严重急性呼吸综合征冠状病毒2的Spike蛋白点突变后与受体蛋白质及潜在抗病毒药物结合能力的同源建模分析[J]. 北京大学学报(医学版), 2021, 53(1): 150-158. |
| [11] | 吴君怡,余淼,孙仕晨,樊壮壮,郑静蕾,张刘陶,冯海兰,刘洋,韩冬. 少汗性外胚层发育不良患者EDA基因突变检测及表型分析[J]. 北京大学学报(医学版), 2021, 53(1): 24-33. |
| [12] | 鲍轶,莫娟芬. 同时性多原发肺腺癌组织编码转录因子ERG基因相同位点突变1例报告[J]. 北京大学学报(医学版), 2020, 52(5): 971-974. |
| [13] | 张宽根,周雨禾,邵雅昆,梅放,由江峰,刘北英,裴斐. 肿瘤转移抑制基因LASS2/TMSG1 S248A突变体通过增加ATP6V0C表达促进前列腺癌的侵袭[J]. 北京大学学报(医学版), 2019, 51(2): 210-220. |
| [14] | 王皓,刘洋,刘浩辰,韩冬,冯海兰. 先天性缺牙患者中BMP2基因突变检测及功能分析[J]. 北京大学学报(医学版), 2019, 51(1): 9-15. |
| [15] | 尤柱,徐丽莉,李雪芬,张建运,杜菁,孙丽莎. 成釉细胞纤维瘤中BRAF突变基因的检测[J]. 北京大学学报(医学版), 2019, 51(1): 4-5. |
|
||