北京大学学报(医学版) ›› 2023, Vol. 55 ›› Issue (6): 975-981. doi: 10.19723/j.issn.1671-167X.2023.06.004
干扰素-α介导系统性红斑狼疮外周血CD56dimCD57+自然杀伤细胞功能的损伤
- 大连医科大学基础医学院免疫学教研室, 辽宁大连 116044
Interferon-α mediating the functional damage of CD56dimCD57+natural killer cells in peripheral blood of systemic lupus erythematosuss
Xiang-ge ZHAO,Jia-qing LIU,Hui-na HUANG,Zhi-min LU,Zi-ran BAI,Xia LI,Jing-jing QI*( )
)
- Department of Immunology, College of Basic Medical Science, Dalian Medical University, Dalian 116044, Liaoning, China
摘要:
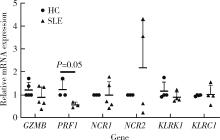
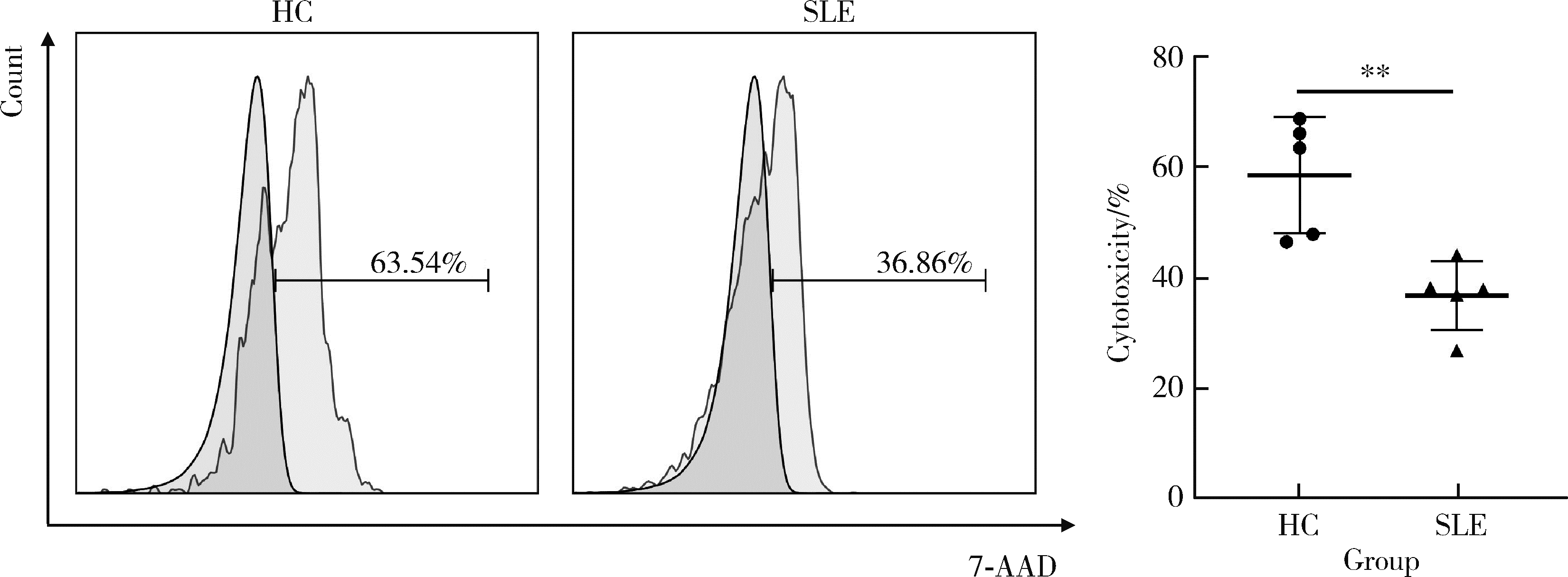
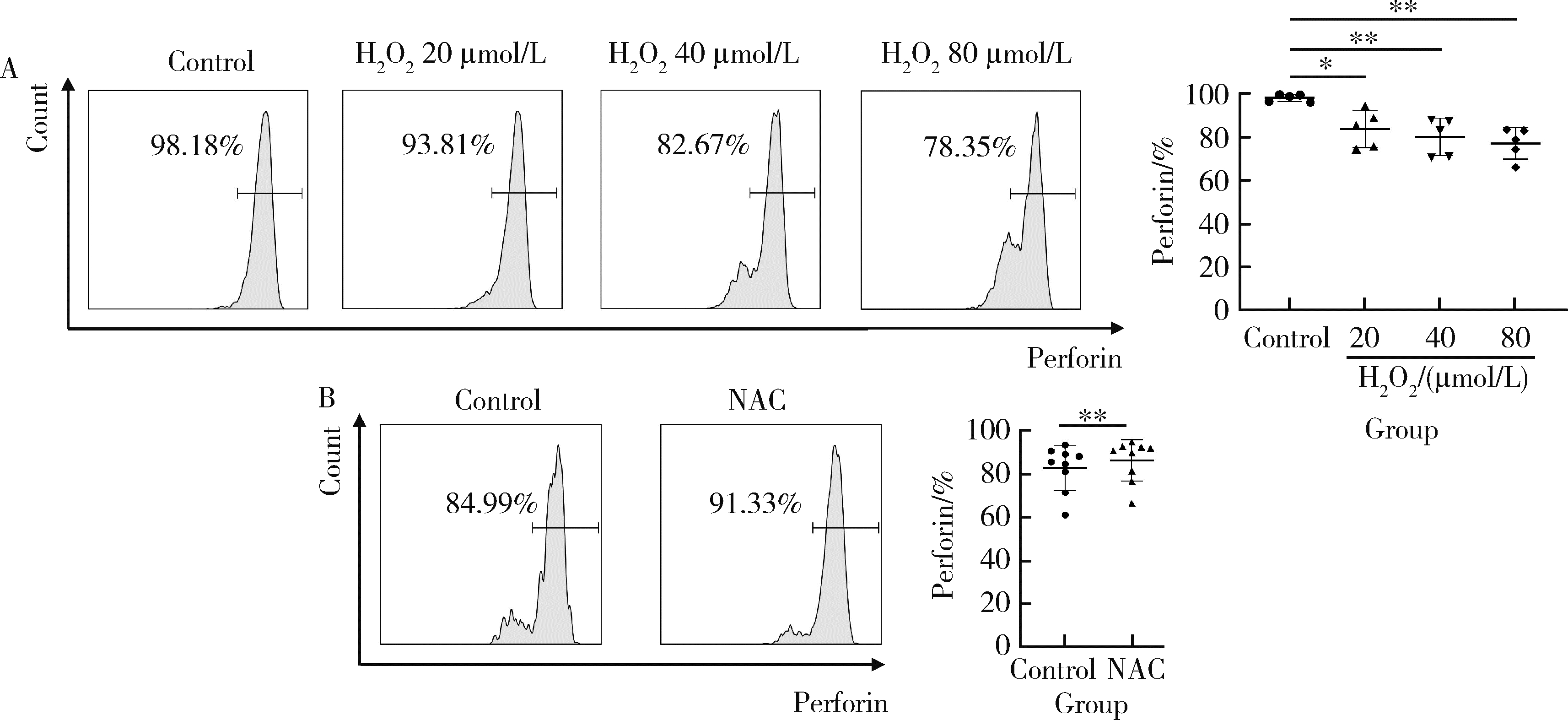
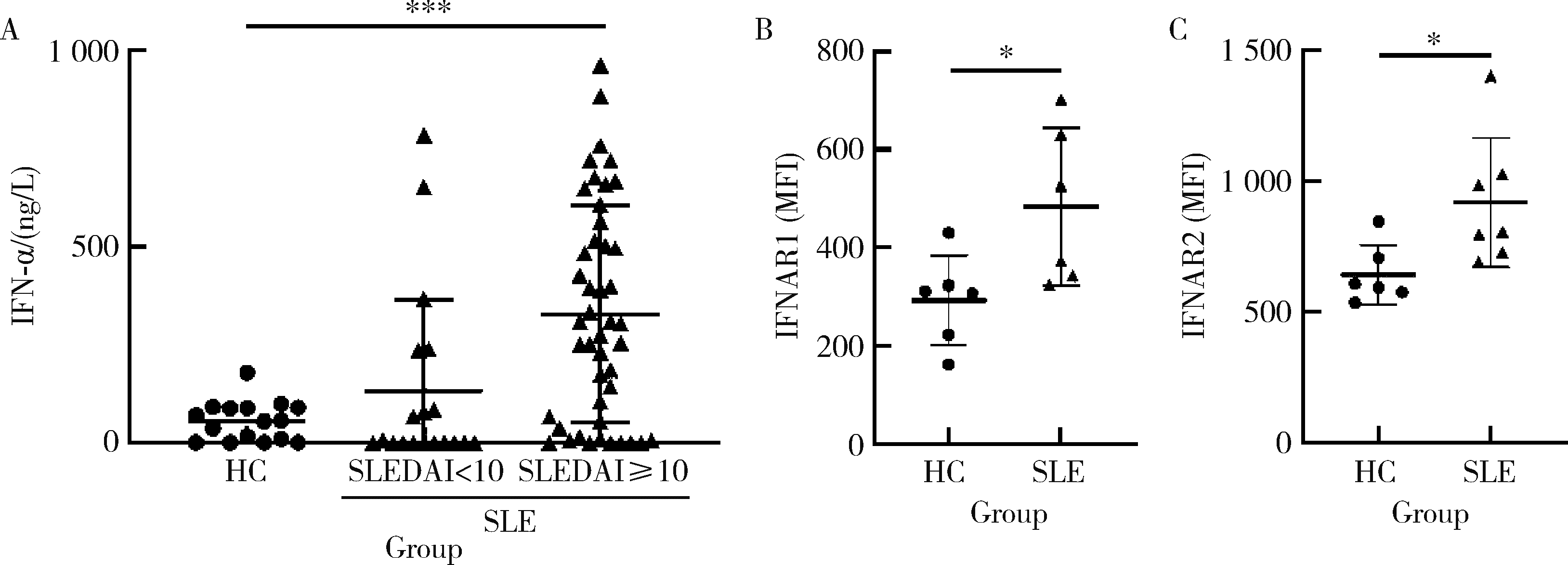
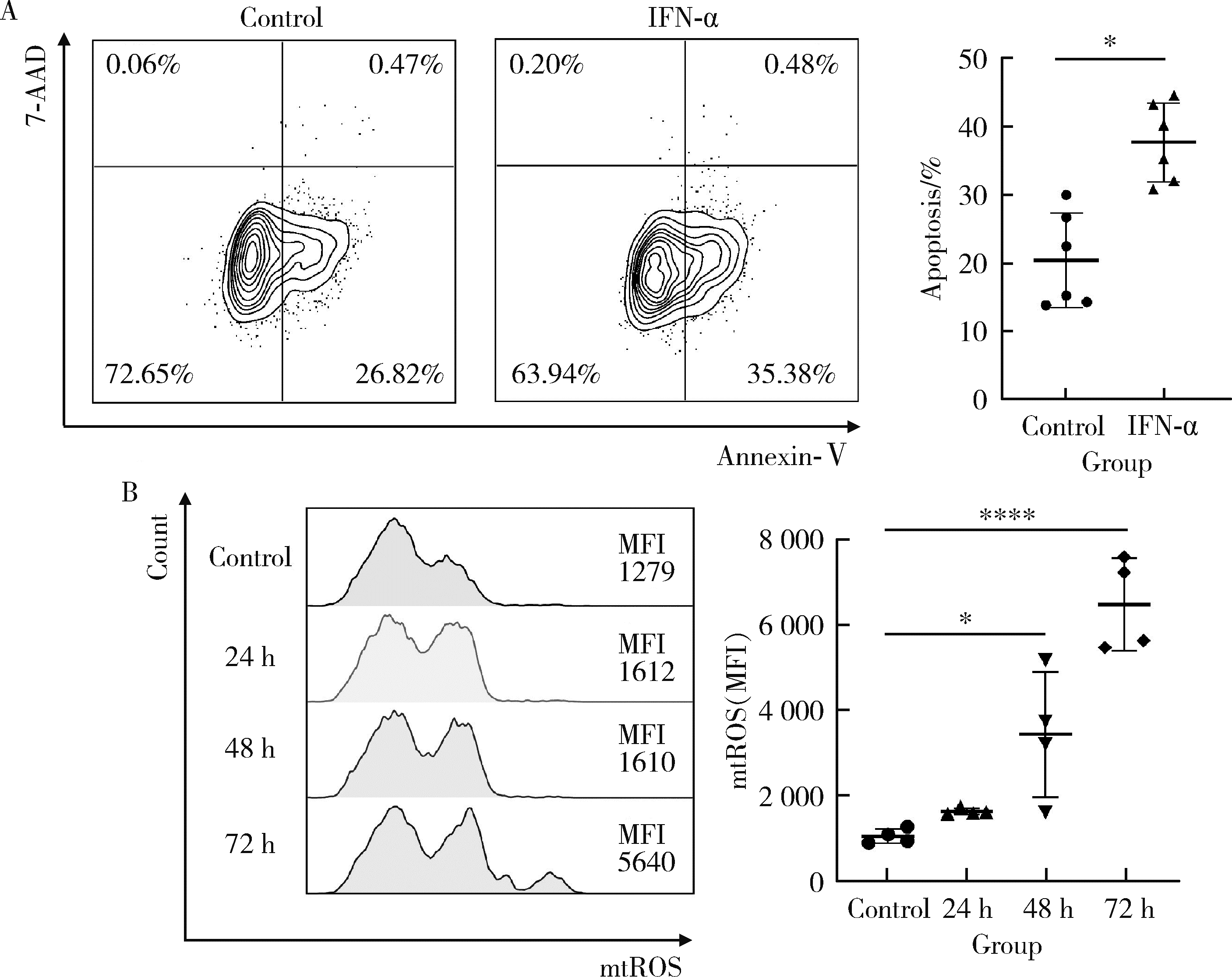
目的: 探究干扰素-α (interferon-α, IFN-α)对系统性红斑狼疮(systemic lupus erythematosus, SLE)患者外周血CD56dimCD57+自然杀伤(natural killer, NK)细胞凋亡和杀伤功能的调控作用及具体机制。方法: 选取大连医科大学附属第二医院就诊的64例初治SLE患者和16例健康人(healthy control, HC)为研究对象,采用实时荧光定量聚合酶链式反应检测NK细胞杀伤功能相关分子的基因表达水平;将CD56dimCD57+NK细胞与K562细胞共培养后,采用膜联蛋白和7氨基放线菌素标记凋亡的K562细胞;用浓度分别为20、40、80 μmol/L的过氧化氢(hydrogen peroxide, H2O2) 处理外周血单个核细胞,并以不使用H2O2处理作为对照组,采用流式细胞术检测穿孔素(per-forin, PRF)表达水平;采用酶联免疫吸附试验测定血清中IFN-α的浓度;采用流式细胞术检测CD56dimCD57+NK细胞表面IFN-α受体(interferon-α receptor, IFNAR)表达水平,并用平均荧光强度(mean fluorescence intensity, MFI)表示;用浓度为1 000 U/mL的IFN-α处理CD56dimCD57+NK细胞24、48、72 h,并以不使用IFN-α处理作为对照组,采用流式细胞术检测细胞凋亡及线粒体活性氧(mitochondrial reactive oxygen species, mtROS)表达水平,并用MFI表示。结果: 与HC(n=3)相比,SLE患者(n=3)外周血总NK细胞的PRF1基因表达水平降低(1.24±0.41 vs. 0.57±0.12, P=0.05)。与HC(n=5)相比,SLE患者(n=5)外周血CD56dimCD57+NK细胞杀伤K562细胞的能力明显下降(58.61%±10.60% vs. 36.74%±6.27%, P < 0.01)。与对照组(n=5, 97.51%±1.67%)相比,不同浓度的H2O2处理可显著下调CD56dimCD57+NK细胞的PRF表达水平且呈剂量依赖性,浓度为20 μmol/L的H2O2组PRF为83.23%±8.48% (n=5, P < 0.05),浓度为40 μmol/L的H2O2组PRF为79.53%±8.56% (n=5, P < 0.01),浓度为80 μmol/L的H2O2组PRF为76.67%±7.16% (n=5,P < 0.01)。与HC (n=16)相比,系统性红斑狼疮疾病活动评分(systemic lupus erythematosus disease activity index, SLEDAI) 为中高疾病活动度(SLEDAI≥10)的SLE患者(n=45)血清IFN-α水平显著增高[(55.07±50.36) ng/L vs. (328.2±276.3) ng/L, P < 0.001]。同时与HC (n=6)相比,SLE患者(n=6)外周血CD56dimCD57+NK细胞IFNAR1表达增高(MFI: 292.7±91.9 vs. 483.2±160.3, P < 0.05),与HC (n=6)相比,SLE患者(n=7)外周血CD56dimCD57+NK细胞IFNAR2表达增高(MFI: 643.5±113.7 vs. 919.0±246.9, P < 0.05)。与对照组(n=6)相比,IFN-α刺激(n=6)可显著促进CD56dimCD57+NK细胞凋亡(20.48%±7.01% vs. 37.82%±5.84%, P < 0.05)。同时,与对照组(n=4, MFI: 1 049±174.5)相比,不同时间下用IFN-α刺激CD56dimCD57+NK细胞可显著促进mtROS的产生,且呈时间依赖性,48 h的MFI为3 437±1 472 (n=4, P < 0.05),72 h的MFI为6 495±1 089 (n=4, P < 0.000 1),而刺激24 h时差异无统计学意义。结论: SLE患者血清中高水平的IFN-α可能通过促进mtROS产生诱导细胞凋亡并抑制PRF表达,下调CD56dimCD57+NK杀伤功能。
中图分类号:
- R593.2
| 1 |
Humbel M , Bellanger F , Fluder N , et al. Restoration of NK cell cytotoxic function with elotuzumab and daratumumab promotes elimination of circulating plasma cells in patients with SLE[J]. Front Immunol, 2021, 12, 645478.
doi: 10.3389/fimmu.2021.645478 |
| 2 |
Cheng Q , Chen X , Xu J , et al. lncRNA X-inactive-specific transcript is a potential biomarker related to changes in CD4+T cell levels in systemic lupus erythematosus[J]. Rheumatol Autoimmun, 2022, 2 (3): 159- 174.
doi: 10.1002/rai2.12048 |
| 3 | Lin SJ , Kuo ML , Hsiao HS , et al. Cytotoxic function and cytokine production of natural killer cells and natural killer T-like cells in systemic lupus erythematosis regulation with interleukin-15[J]. Mediators Inflamm, 2019, 2019, 4236562. |
| 4 |
Sordo-Bahamonde C , Lorenzo-Herrero S , Payer ÁR , et al. Mechanisms of apoptosis resistance to NK cell-mediated cytotoxicity in cancer[J]. Int J Mol Sci, 2020, 21 (10): 3726.
doi: 10.3390/ijms21103726 |
| 5 | Lorenzo-Herrero S , Sordo-Bahamonde C , Gonzalez S , et al. CD107a degranulation assay to evaluate immune cell antitumor activity[J]. Methods Mol Biol, 2019, 1884, 119- 130. |
| 6 |
Myers JA , Miller JS . Exploring the NK cell platform for cancer immunotherapy[J]. Nat Rev Clin Oncol, 2021, 18 (2): 85- 100.
doi: 10.1038/s41571-020-0426-7 |
| 7 |
Quatrini L , Della Chiesa M , Sivori S , et al. Human NK cells, their receptors and function[J]. Eur J Immunol, 2021, 51 (7): 1566- 1579.
doi: 10.1002/eji.202049028 |
| 8 |
Kucuksezer UC , Aktas Cetin E , Esen F , et al. The role of natural killer cells in autoimmune diseases[J]. Front Immunol, 2021, 12, 622306.
doi: 10.3389/fimmu.2021.622306 |
| 9 |
Bryceson YT , March ME , Barber DF , et al. Cytolytic granule polarization and degranulation controlled by different receptors in resting NK cells[J]. J Exp Med, 2005, 202 (7): 1001- 1012.
doi: 10.1084/jem.20051143 |
| 10 | Nielsen CM , White MJ , Goodier MR , et al. Functional significance of CD57 expression on human NK cells and relevance to disease[J]. Front Immunol, 2013, 4, 422. |
| 11 |
Lu Z , Tian Y , Bai Z , et al. Increased oxidative stress contributes to impaired peripheral CD56dimCD57+ NK cells from patients with systemic lupus erythematosus[J]. Arthritis Res Ther, 2022, 24 (1): 48.
doi: 10.1186/s13075-022-02731-y |
| 12 |
Banchereau R , Hong S , Cantarel B , et al. Personalized immunomonitoring uncovers molecular networks that stratify lupus patients[J]. Cell, 2016, 165 (3): 551- 565.
doi: 10.1016/j.cell.2016.03.008 |
| 13 |
Li P , Jiang M , Li K , et al. Glutathione peroxidase 4-regulated neutrophil ferroptosis induces systemic autoimmunity[J]. Nat Immunol, 2021, 22 (9): 1107- 1117.
doi: 10.1038/s41590-021-00993-3 |
| 14 |
Furie R , Werth VP , Merola JF , et al. Monoclonal antibody targeting BDCA2 ameliorates skin lesions in systemic lupus erythematosus[J]. J Clin Invest, 2019, 129 (3): 1359- 1371.
doi: 10.1172/JCI124466 |
| 15 |
Chatham WW , Furie R , Saxena A , et al. Long-term safety and efficacy of anifrolumab in adults with systemic lupus erythematosus: Results of a phase Ⅱ open-label extension study[J]. Arthritis Rheumatol, 2021, 73 (5): 816- 825.
doi: 10.1002/art.41598 |
| 16 |
Kirou KA , Mavragani CP , Crow MK . Activation of type I inter-feron in systemic lupus erythematosus[J]. Expert Rev Clin Immunol, 2007, 3 (4): 579- 588.
doi: 10.1586/1744666X.3.4.579 |
| 17 |
Le Bon A , Thompson C , Kamphuis E , et al. Cutting edge: Enhancement of antibody responses through direct stimulation of B and T cells by type I IFN[J]. J Immunol, 2006, 176 (4): 2074- 2078.
doi: 10.4049/jimmunol.176.4.2074 |
| 18 |
Cucak H , Yrlid U , Reizis B , et al. Type Ⅰ interferon signaling in dendritic cells stimulates the development of lymph-node-resident T follicular helper cells[J]. Immunity, 2009, 31 (3): 491- 501.
doi: 10.1016/j.immuni.2009.07.005 |
| 19 | De Groof A , Ducreux J , Aleva F , et al. STAT3 phosphorylation mediates the stimulatory effects of interferon alpha on B cell dif-ferentiation and activation in SLE[J]. Rheumatology (Oxford), 2020, 59 (3): 668- 677. |
| 20 | Hochberg MC . Updating the American College of Rheumatology revised criteria for the classification of systemic lupus erythematosus[J]. Arthritis Rheum, 1997, 40 (9): 1725. |
| 21 | Li H , Geng L , Cao Z , et al. CD56brightCD16- to CD57+CD56dimCD16+ NK cell ratio discriminates disease activity and renal involvement in patients with systemic lupus erythematosus[J]. Clin Exp Rheumatol, 2023, 41 (9): 1768- 1776. |
| 22 |
Yang Y , Day J , Souza-Fonseca Guimaraes F , et al. Natural killer cells in inflammatory autoimmune diseases[J]. Clin Transl Immunology, 2021, 10 (2): e1250.
doi: 10.1002/cti2.1250 |
| 23 |
Streltsova MA , Erokhina SA , Kanevskiy LM , et al. Analysis of NK cell clones obtained using interleukin-2 and gene-modified K562 cells revealed the ability of "senescent" NK cells to lose CD57 expression and start expressing NKG2A[J]. PLoS One, 2018, 13 (12): e0208469.
doi: 10.1371/journal.pone.0208469 |
| 24 | Bahadorian D , Mollazadeh S , Mirazi H , et al. Regulatory NK cells in autoimmune disease[J]. Iran J Basic Med Sci, 2023, 26 (6): 609- 616. |
| 25 |
Fresneda Alarcon M , McLaren Z , Wright HL . Neutrophils in the pathogenesis of rheumatoid arthritis and systemic lupus erythematosus: Same foe different M.O[J]. Front Immunol, 2021, 12, 649693.
doi: 10.3389/fimmu.2021.649693 |
| 26 |
Shah D , Mahajan N , Sah S , et al. Oxidative stress and its biomarkers in systemic lupus erythematosus[J]. J Biomed Sci, 2014, 21 (1): 23.
doi: 10.1186/1423-0127-21-23 |
| 27 |
Kennel KB , Greten FR . Immune cell-produced ROS and their impact on tumor growth and metastasis[J]. Redox Biol, 2021, 42, 101891.
doi: 10.1016/j.redox.2021.101891 |
| 28 |
Song H , Park H , Kim YS , et al. L-kynurenine-induced apoptosis in human NK cells is mediated by reactive oxygen species[J]. Int Immunopharmacol, 2011, 11 (8): 932- 938.
doi: 10.1016/j.intimp.2011.02.005 |
| 29 |
He H , Wang C , Liu G , et al. Isobavachalcone inhibits acute myeloid leukemia: Potential role for ROS-dependent mitochondrial apoptosis and differentiation[J]. Phytother Res, 2021, 35 (6): 3337- 3350.
doi: 10.1002/ptr.7054 |
| 30 |
Bhattacharya A , Hegazy AN , Deigendesch N , et al. Superoxide dismutase 1 protects hepatocytes from type I interferon-driven oxidative damage[J]. Immunity, 2015, 43 (5): 974- 986.
doi: 10.1016/j.immuni.2015.10.013 |
| 31 |
Tasdogan A , Kumar S , Allies G , et al. DNA damage-induced HSPC malfunction depends on ROS accumulation downstream of IFN-1 signaling and bid mobilization[J]. Cell Stem Cell, 2016, 19 (6): 752- 767.
doi: 10.1016/j.stem.2016.08.007 |
| 32 |
Buang N , Tapeng L , Gray V , et al. Type Ⅰ interferons affect the metabolic fitness of CD8+ T cells from patients with systemic lupus erythematosus[J]. Nat Commun, 2021, 12 (1): 980.
doi: 10.1038/s41467-021-21210-7 |
| [1] | 汤晓菲, 丁秋玲, 张阳, 王育梅, 田美伊, 孙卓, 王荣荣, 陈妍, 刘坚. 阿伐曲泊帕治疗难治性系统性红斑狼疮相关血小板减少1例[J]. 北京大学学报(医学版), 2026, 58(2): 405-409. |
| [2] | 郑苗, 马欣蓉, 陈昊, 赵恒欣, 张宇, 谭建国, 李和平, 王霄. 大气压放电冷等离子体处理对人牙龈成纤维细胞生物学行为的影响[J]. 北京大学学报(医学版), 2026, 58(1): 60-67. |
| [3] | 李欣艺, 赵金霞, 穆荣. 阿司匹林对系统性红斑狼疮妊娠者结局的影响[J]. 北京大学学报(医学版), 2025, 57(6): 1074-1080. |
| [4] | 王晓林, 郭邵逸, 陈大召, 温锡杰, 华勇, 张亮, 张秦. 全髋关节置换术治疗系统性红斑狼疮继发股骨头缺血性坏死的随访研究[J]. 北京大学学报(医学版), 2025, 57(6): 1081-1088. |
| [5] | 卫春, 杨月, 赵新菊, 刘栩, 贾园. 系统性红斑狼疮合并自身免疫性郎飞结病1例[J]. 北京大学学报(医学版), 2025, 57(6): 1174-1179. |
| [6] | 赵凯, 鲁芙爱, 王永福. 中枢神经系统感染模拟神经精神狼疮1例[J]. 北京大学学报(医学版), 2025, 57(6): 1188-1192. |
| [7] | 王小梦, 曾晓君, 李娟. 黎族与汉族系统性红斑狼疮患者的主要临床特征[J]. 北京大学学报(医学版), 2025, 57(6): 1213-1218. |
| [8] | 王文琼, 侯玉珂, 李春, 张学武. 系统性红斑狼疮患者不良妊娠结局的预测因素[J]. 北京大学学报(医学版), 2025, 57(3): 599-603. |
| [9] | 王红彦, 李鑫铭, 房柯池, 朱华群, 贾汝琳, 王晶. 系统性红斑狼疮疾病活动度相关特征分析及评估模型的构建[J]. 北京大学学报(医学版), 2024, 56(6): 1017-1022. |
| [10] | 陈丹丹, 李云, 卢情怡, 相晓红, 孙峰, 李英妮, 赵静, 王红彦, 李春. 育龄期系统性红斑狼疮患者卵巢功能的评价及其影响因素[J]. 北京大学学报(医学版), 2024, 56(6): 1023-1028. |
| [11] | 王莉, 高超, 任欢欢, 沈艳平, 黄晓玮, 姚鸿, 韩丹丹. 系统性红斑狼疮患者自我管理能力现状及相关因素分析[J]. 北京大学学报(医学版), 2024, 56(6): 1029-1035. |
| [12] | 柴静, 王钥, 穆荣, 赵金霞. 系统性红斑狼疮累及穹窿柱导致低钠血症1例[J]. 北京大学学报(医学版), 2024, 56(6): 1115-1118. |
| [13] | 王明霞, 丁菱, 王敏, 邹婵娟, 颜丝语, 梁颖文, 王伟佳, 何善智. 双靶点嵌合抗原受体T细胞治疗系统性红斑狼疮患者停药后安全孕产1例[J]. 北京大学学报(医学版), 2024, 56(6): 1119-1125. |
| [14] | 田佳宜, 郭一学, 张霞, 孙晓麟, 何菁. 中国健康成人外周血自然杀伤细胞及其亚群的正常值范围流式细胞学分析[J]. 北京大学学报(医学版), 2024, 56(5): 839-844. |
| [15] | 武志慧, 胡明智, 赵巧英, 吕凤凤, 张晶莹, 张伟, 王永福, 孙晓林, 王慧. miR-125b-5p修饰脐带间充质干细胞对系统性红斑狼疮的免疫调控机制[J]. 北京大学学报(医学版), 2024, 56(5): 860-867. |
|
||