北京大学学报(医学版) ›› 2022, Vol. 54 ›› Issue (5): 971-980. doi: 10.19723/j.issn.1671-167X.2022.05.026
阿替利珠单抗治疗中国晚期实体瘤患者的开放标签Ⅰ期临床试验
张力1,龚继芳2,潘宏铭3,白玉贤4,刘天舒5,程颖6,陈亚池7,黄佳莹8,许婷婷8,葛飞娇8,许婉玲9,施佳10,胡夕春11,*( ),沈琳2,*(
),沈琳2,*( )
)
- 1. 中山大学附属肿瘤医院肿瘤内科, 广州 510060
2. 北京大学肿瘤医院暨北京市肿瘤防治研究所消化肿瘤内科, 恶性肿瘤发病机制及转化研究教育部重点实验室, 北京 100142
3. 浙江大学医学院附属邵逸夫医院肿瘤内科, 杭州 310020
4. 哈尔滨医科大学附属肿瘤医院肿瘤内科, 哈尔滨 150081
5. 复旦大学附属中山医院肿瘤内科, 上海 200032
6. 吉林省肿瘤医院肿瘤内科, 长春 130012
7. Clinical Pharmacology, Genentech, Inc., South San Francisco, CA 94080, USA
8. 罗氏全球药品开发中国中心临床科学部, 上海 201203
9. 罗氏全球药品开发中国中心统计部, 上海 201203
10. 罗氏全球药品开发中国中心安全部, 上海 201203
11. 复旦大学附属肿瘤医院肿瘤内科, 上海 200032
Atezolizumab therapy in Chinese patients with locally advanced or metastatic solid tumors: An open-label, phase Ⅰ study
Li ZHANG1,Ji-fang GONG2,Hong-ming PAN3,Yu-xian BAI4,Tian-shu LIU5,Ying CHENG6,Ya-chi CHEN7,Jia-ying HUANG8,Ting-ting XU8,Fei-jiao GE8,Wan-ling HSU9,Jane SHI10,Xi-chun HU11,*( ),Lin SHEN2,*(
),Lin SHEN2,*( )
)
- 1. Department of Medical Oncology, Sun Yat-sen University Cancer Center, Guangzhou 510060, China
2. Key Laboratory of Carcinogenesis and Translational Research, Ministry of Education; Department of Gastrointestinal Oncology, Peking University Cancer Hospital & Institute, Beijing 100142, China
3. Department of Medical Oncology, Sir Run Run Shaw Hospital, Zhejiang University School of Medicine, Hangzhou 310020, China
4. Department of Medical Oncology, Harbin Medical University Cancer Hospital, Harbin 150081, China
5. Department of Medical Oncology, Zhongshan Hospital, Fudan University, Shanghai 200032, China
6. Department of Medical Oncology, Jilin Cancer Hospital, Changchun 130012, China
7. Clinical Pharmacology, Genentech, Inc., South San Francisco, CA 94080, USA
8. Oncology, Roche Product Development Shanghai, Shanghai, 201203, China
9. Department of Statistics, Roche Product Development Shanghai, Shanghai 201203, China
10. Portfolio Clinical Safety, Roche Product Development Shanghai, Shanghai 201203, China
11. Department of Medical Oncology, Fudan University Shanghai Cancer Center, Shanghai 200032, China
摘要:
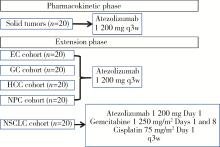
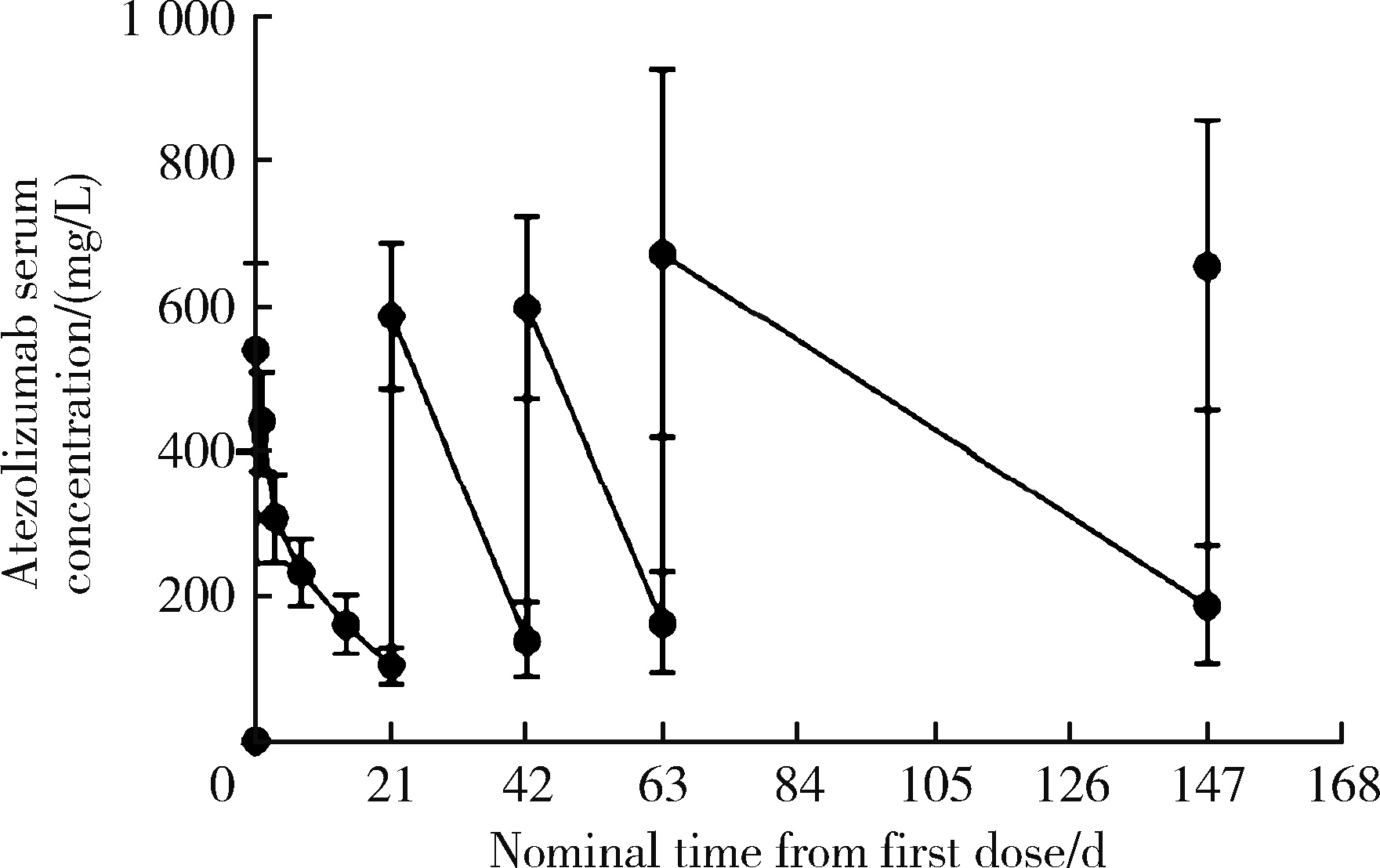
目的: 观察程序性细胞死亡配体1(programmed death-ligand 1,PD-L1)抑制剂阿替利珠单抗在中国高发实体瘤,包括食管癌(esophageal cancer,EC)、胃癌(gastric cancer,GC)、肝细胞癌(hepatocellular carcinoma,HCC)、鼻咽癌(nasopharyngeal cancer,NPC)和非小细胞肺癌(non-small cell lung cancer,NSCLC)患者中的药代动力学(pharmacokinetics,PK)、疗效和安全性数据。方法: 本研究为开放标签的Ⅰ期临床试验,于2016年8月4日至2019年4月15日在中国6个研究中心进行。入组患者年龄≥18岁,患有经组织学证实的无法治愈或转移性的实体瘤,且既往抗肿瘤治疗失败。PK阶段研究了阿替利珠单抗单药治疗的PK和安全性;扩展阶段研究了阿替利珠单抗单药治疗(入组EC、GC、HCC、NPC患者)和联合化疗(入组NSCLC患者)的安全性和有效性。结果: 共入组120例患者(PK阶段20例;扩展期每队列20例)。阿替利珠单抗单药组患者(n=100)中有42例(42.0%)为PD-L1阳性,9例(9.0%)为微卫星高度不稳定性。阿替利珠单抗的清除率为0.219 L/d,重复给药6~9周(2~3个周期)后达到稳态。EC、GC、HCC、NPC和NSCLC的客观缓解率(objective response rate,ORR)分别为10.0%、15.0%、10.0%、5.0%和40.0%。在PD-L1阳性的肿瘤患者中,阿替利珠单抗的ORR为11.9%,阿替利珠单抗联合吉西他滨和顺铂的ORR为46.2%。2例GC患者在假性进展后获得了持久的肿瘤缩小。阿替利珠单抗单药组最常见的治疗相关不良事件是疲劳、贫血、发热和白细胞计数减少,联合组最常见的治疗相关AE是贫血、白细胞计数减少和食欲下降。本试验没有发现新的安全信号。结论: 中国患者应用阿替利珠单抗的PK、疗效和安全性与之前研究中入组的全球患者的数据相似。
中图分类号:
- R730.51
| 1 | International Agency for Research on Cancer. China (Globocan 2020)[EB/OL]. (2021-03)[2021-05-15] https://gco.iarc.fr/today/data/factsheets/populations/160-china-fact-sheets.pdf. |
| 2 |
Arnold M , Soerjomataram I , Ferlay J , et al. Global incidence of oesophageal cancer by histological subtype in 2012[J]. Gut, 2015, 64 (3): 381- 387.
doi: 10.1136/gutjnl-2014-308124 |
| 3 | Salehiniya H , Mohammadian M , Mohammadian-Hafshejani A , et al. Nasopharyngeal cancer in the world: Epidemiology, incidence, mortality and risk factors[J]. World Cancer Res J, 2018, 5 (1): e1046. |
| 4 | Merck. Highlights of prescribing information. Keytruda® (Pembrolizumab)[EB/OL]. (2014)[2022-05-12]. https://www.merck.com/product/usa/pi_circulars/k/keytruda/keytruda_pi.pdf. |
| 5 |
Fuchs CS , Doi T , Jang RW , et al. Safety and efficacy of pembro-lizumab monotherapy in patients with previously treated advanced gastric and gastroesophageal junction cancer: phase 2 clinical KEYNOTE-059 trial[J]. JAMA Oncol, 2018, 4 (5): e180013.
doi: 10.1001/jamaoncol.2018.0013 |
| 6 | Bristol Myers Squibb Company. Highlights of prescribing information. Opdivo (Nivolumab)[EB/OL]. (2014)[2022-05-03]. https://packageinserts.bms.com/pi/pi_opdivo.pdf. |
| 7 |
Ma BBY , Lim WT , Goh BC , et al. Antitumor activity of ni-volumab in recurrent and metastatic nasopharyngeal carcinoma: An international, multicenter study of the Mayo clinic phase 2 consortium (NCI-9742)[J]. J Clin Oncol, 2018, 36 (14): 1412- 1418.
doi: 10.1200/JCO.2017.77.0388 |
| 8 | NCCN Clinical Practice Guidelines in Oncology. Non-small cell lung cancer. Version 3.2022[EB/OL]. (2022-03-16)[2022-05]. http://www.nccn.org/professionals/physician_gls/pdf/nscl.pdf. |
| 9 |
Herbst RS , Soria JC , Kowanetz M , et al. Predictive correlates of response to the anti-PD-L1 antibody MPDL3280A in cancer patients[J]. Nature, 2014, 515 (7528): 563- 567.
doi: 10.1038/nature14011 |
| 10 |
Weinstock C , Khozin S , Suzman D , et al. U.S. Food and Drug Administration approval summary: Atezolizumab for metastatic non-small cell lung cance[J]. Clin Cancer Res, 2017, 23 (16): 4534- 4539.
doi: 10.1158/1078-0432.CCR-17-0540 |
| 11 | Genentech. FDA approves Genentech's Tecentriq plus chemothe-rapy (abraxane and carboplatin) for the initial treatment of metastatic non-squamous non-small cell lung cancer[N/OL]. (2019-12-03)[2021-05-03] https://www.gene.com/media/press-releases/14827/2019-12-03/fda-approves-genentechs-tecentriq-plus-c. |
| 12 | F. Hoffmann-La Roche Ltd. European Commission approves Roche's new Tecentriq-based combination therapy as an initial treatment for most common form of advanced lung cancer[N/OL]. (2019-09-06)[2021-05-11] https://www.roche.com/media/releases/med-cor-2019-09-06.htm. |
| 13 | F. Hoffmann-La Roche Ltd. FDA approves Roche's Tecentriq in combination with Avastin for people with the most common form of liver cancer[N/OL]. (2020-06-02)[2021-05-15] https://www.roche.com/media/releases/med-cor-2020-06-02.htm. |
| 14 |
Finn RS , Qin S , Ikeda M , et al. Atezolizumab plus Bevacizumab in unresectable hepatocellular carcinoma[J]. N Engl J Med, 2020, 382 (20): 1894- 1905.
doi: 10.1056/NEJMoa1915745 |
| 15 | Xu ZY , Brown L , Pan GW , et al. Lifestyle, environmental pollution and lung cancer in cities of Liaoning in northeastern China[J]. Lung Cancer, 1996, 14 (Suppl 1): S149- S160. |
| 16 |
Grenade C , Phelps MA , Villalona-Calero MA . Race and ethnicity in cancer therapy: What have we learned?[J]. Clin Pharmacol Ther, 2014, 95 (4): 403- 412.
doi: 10.1038/clpt.2014.5 |
| 17 | Genentech. Highlights of prescribing information. Tecentriq®(atezolizumab)[EB/OL]. (2016)[2022-05-15]. https://www.gene.com/download/pdf/tecentriq_prescribing.pdf. |
| 18 |
West H , McCleod M , Hussein M , et al. Atezolizumab in combination with carboplatin plus nab-paclitaxel chemotherapy compared with chemotherapy alone as first-line treatment for metastatic non-squamous non-small-cell lung cancer (IMpower130): A multicentre, randomised, open-label, phase 3 trial[J]. Lancet Oncol, 2019, 20 (7): 924- 937.
doi: 10.1016/S1470-2045(19)30167-6 |
| 19 |
Lee MS , Ryoo BY , Hsu CH , et al. Atezolizumab with or without bevacizumab in unresectable hepatocellular carcinoma (GO30140): An open-label, multicentre, phase 1b study[J]. Lancet Oncol, 2020, 21 (6): 808- 820.
doi: 10.1016/S1470-2045(20)30156-X |
| 20 |
Zhang T , Xie J , Arai S , et al. The efficacy and safety of anti-PD-1/PD-L1 antibodies for treatment of advanced or refractory can-cers: A meta-analysis[J]. Oncotarget, 2016, 7 (45): 73068- 73079.
doi: 10.18632/oncotarget.12230 |
| 21 |
Le DT , Kavan P , Kim TW , et al. KEYNOTE-164: Pembrolizu-mab for patients with advanced microsatellite instability high (MSI-H) colorectal cancer[J]. J Clin Oncol, 2018, 36 (Suppl 15): 3514.
doi: 10.1200/jco.2018.36.15_suppl.3514 |
| 22 |
Colevas AD , Bahleda R , Braiteh F , et al. Safety and clinical activity of atezolizumab in head and neck cancer: Results from a phase Ⅰ trial[J]. Ann Oncol, 2018, 29 (11): 2247- 2253.
doi: 10.1093/annonc/mdy411 |
| 23 |
Schiller JH , Harrington D , Belani CP , et al. Comparison of four chemotherapy regimens for advanced non-small-cell lung cancer[J]. N Engl J Med, 2002, 346 (2): 92- 98.
doi: 10.1056/NEJMoa011954 |
| 24 | International Agency for Research on Cancer. All cancers (Globocan 2020)[EB/OL]. (2020-12)[2022-05-12] https://gco.iarc.fr/today/data/factsheets/cancers/39-All-cancers-fact-sheet.pdf. |
| [1] | 季加孚, 韦静涛, 季科, 步召德. 胃癌诊疗的瓶颈与破局:迈向精准化与智能化融合的新纪元[J]. 北京大学学报(医学版), 2026, 58(2): 231-238. |
| [2] | 高加勒, 张忠涛. 局部进展期直肠癌精准治疗现状与展望[J]. 北京大学学报(医学版), 2026, 58(2): 247-250. |
| [3] | 王海, 江一舟. 靶向血管治疗在乳腺癌精准治疗中的分子机制与临床应用[J]. 北京大学学报(医学版), 2026, 58(2): 251-256. |
| [4] | 罗必显, 刘洪铭, 谢伟勋, 龚渭华. 产甲胎蛋白胃癌的新临床特征和前沿科学问题[J]. 北京大学学报(医学版), 2026, 58(2): 257-265. |
| [5] | 杜文, 章文博, 于尧, 刘硕, 苏惠裕, 胡耒豪, 唐祖南, 吴彬彰, 陈震, 李家琦, 王昊, 彭歆. 口腔颌面部肿瘤"数智化外科"诊疗流程探索与临床应用[J]. 北京大学学报(医学版), 2026, 58(2): 278-284. |
| [6] | 王楠楠, 袁大晋, 朱昱冰, 丁磊. 结直肠癌根治术后肝转移风险多中心列线图预测模型的构建与验证[J]. 北京大学学报(医学版), 2026, 58(2): 290-300. |
| [7] | 刘友东, 吕亚军, 陈杰, 臧明德, 潘宏达, 刘晓文, 陆俊, 刘凤林. 全腹腔镜保留贲门胃底胃次全切除术治疗中上部胃癌的疗效及安全性[J]. 北京大学学报(医学版), 2026, 58(2): 301-306. |
| [8] | 李嘉临, 陈力侨, 唐家天, 吴艳, 王安强. 胃肝样腺癌转化治疗1例[J]. 北京大学学报(医学版), 2026, 58(2): 399-404. |
| [9] | 李斌, 梁寒. 机器人胃癌根治术:研究进展与实践挑战[J]. 北京大学学报(医学版), 2026, 58(2): 416-422. |
| [10] | 董海峰, 陈恒星, 张常华. 恶性肿瘤中蛋白质乳酸化修饰的研究进展[J]. 北京大学学报(医学版), 2026, 58(2): 423-430. |
| [11] | 李宏杨, 黄涛, 王琳琳. 脂肪肌肉比率与卵巢良性肿瘤风险的关联性[J]. 北京大学学报(医学版), 2026, 58(1): 169-174. |
| [12] | 高若凡, 马天宇, 王润楷, 殷雨辰, 李芮迪, 王丹丹, 夏斌. 细胞膜囊泡递送靶向肿瘤坏死因子-α的小干扰RNA对牙髓干细胞的抗炎作用[J]. 北京大学学报(医学版), 2026, 58(1): 22-29. |
| [13] | 刘艳华, 陆敏, 赵旭阳, 张宽根, 武睿, 梅放, 戴志豪, 由江峰, 裴斐. 肿瘤转移抑制基因LASS2去磷酸化对液泡型ATP酶活性及前列腺癌侵袭性的影响[J]. 北京大学学报(医学版), 2025, 57(6): 1113-1123. |
| [14] | 杨小勇, 张帆, 马潞林, 刘承. 前列腺导管腺癌临床特征及腺外侵犯的影响因素[J]. 北京大学学报(医学版), 2025, 57(5): 956-960. |
| [15] | 陈定一, 杜浩鑫, 张逸晨, 王闫飞, 刘巍, 焦园园, 史录文, 管晓东, 卢新璞. 姑息治疗对晚期癌症患者药物使用和医疗资源利用的影响[J]. 北京大学学报(医学版), 2025, 57(5): 996-1001. |
|
||