北京大学学报(医学版) ›› 2021, Vol. 53 ›› Issue (6): 1083-1087. doi: 10.19723/j.issn.1671-167X.2021.06.013
系统性红斑狼疮患者血清白细胞介素-2受体α水平及其临床意义
- 北京大学人民医院风湿免疫科,北京 100044
Serum interleukin-2 receptor α as a clinical biomarker in patients with systemic lupus erythematosus
TIAN Jia-yi,ZHANG Xia( ),CHENG Gong,LIU Qing-hong,WANG Shi-yang,HE Jing(
),CHENG Gong,LIU Qing-hong,WANG Shi-yang,HE Jing( )
)
- Department of Rheumatology and Immunology, Peking University People’s Hospital, Beijing 100044, China
摘要:
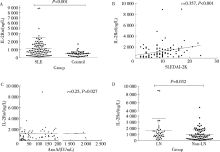
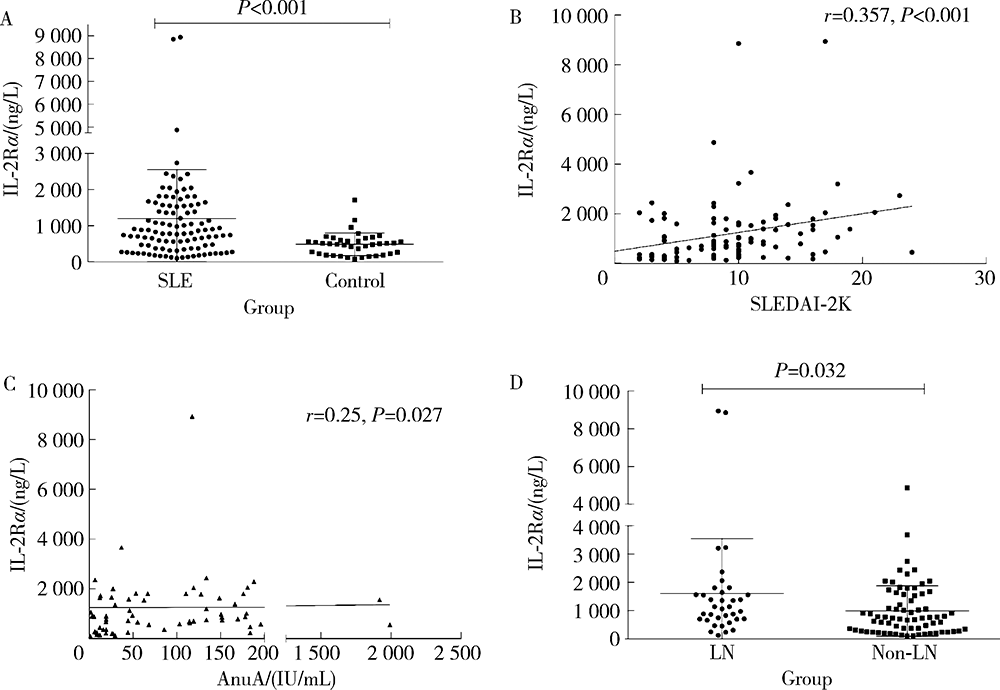
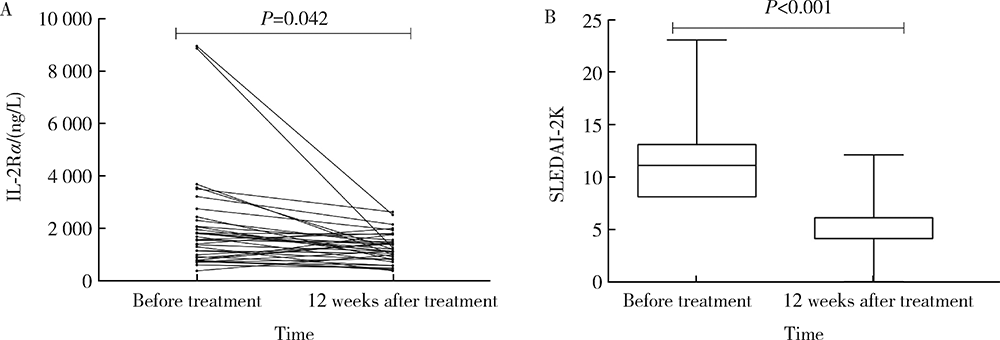
目的:探究系统性红斑狼疮(systemic lupus erythematosus,SLE)患者血清中白细胞介素-2受体α(interleukin-2 receptor α,IL-2Rα)水平在临床中的意义。方法:收集2019年1月至2020年12月就诊于北京大学人民医院的107例SLE患者病历资料,依据SLE疾病活动度指数(SLE disease activity index 2000,SLEDAI-2K)评估患者的病情活动情况,并选取年龄、性别分别匹配的39例健康人作为健康对照。采用酶联免疫吸附法测定SLE患者组和健康对照组的血清IL-2Rα水平,比较其差异并分析SLE患者IL-2Rα水平与临床指标及实验室指标的相关性。采用t检验或Mann-Whitney U检验、 χ2检验和Spearman秩相关性分析进行统计学分析。结果:SLE患者血清IL-2Rα水平[830.82(104.2~8 940.48) ng/L]较健康对照组[505.1(78.65~1 711.52) ng/L]明显升高(P<0.001)。相关性分析显示,血清IL-2Rα水平与SLEDAI-2K评分及抗核小体抗体滴度呈正相关(r=0.357,P<0.001;r=0.25,P=0.027)。107例SLE患者中36例(33.6%)合并狼疮性肾炎,合并狼疮性肾炎的患者血清IL-2Rα水平[1 102.14(126.52~8 940.48) ng/L]较未合并狼疮性肾炎患者[743.89(104.19~4 872.06) ng/L]明显升高(P=0.032)。高IL-2Rα水平组合并狼疮性肾炎者(40.8%)较低水平组(19.4%)明显升高(P=0.031),高IL-2Rα水平组SLEDAI-2K评分更高[10 (3~21) vs. 7 (3~16),P=0.001]。SLE患者常规治疗12周后血清IL-2Rα水平[1 119.1(372.25~2 608.86) ng/L]随病情改善较基线时[1 556.73 (373.08~8 940.48) ng/L]明显下降(P=0.042)。结论:血清IL-2Rα可作为SLE病情活动评估指标,与肾脏受累有一定相关性。
中图分类号:
- R593.24
| [1] |
Tsokos GC. Autoimmunity and organ damage in systemic lupus erythematosus[J]. Nat Immunol, 2020, 21(6):605-614.
doi: 10.1038/s41590-020-0677-6 |
| [2] |
Ross SH, Cantrell DA. Signaling and function of interleukin-2 in T lymphocytes[J]. Annu Rev Immunol, 2018, 36:411-433.
doi: 10.1146/immunol.2018.36.issue-1 |
| [3] | He J, Zhang R, Shao M, et al. Efficacy and safety of low-dose IL-2 in the treatment of systemic lupus erythematosus: A randomised, double-blind, placebo-controlled trial[J]. Ann Rheum Dis, 2020, 79(1):141-149. |
| [4] |
He J, Zhang X, Wei Y, et al. Low-dose interleukin-2 treatment selectively modulates CD4(+) T cell subsets in patients with systemic lupus erythematosus[J]. Nat Med, 2016, 22(9):991-993.
doi: 10.1038/nm.4148 |
| [5] |
Rubin L A, Galli F, Greene WC, et al. The molecular basis for the generation of the human soluble interleukin 2 receptor[J]. Cytokine, 1990, 2(5):330-336.
pmid: 2103332 |
| [6] | Dik WA, Heron M. Clinical significance of soluble interleukin-2 receptor measurement in immune-mediated diseases[J]. Neth J Med, 2020, 78(5):220-231. |
| [7] |
Luo H, Wang C, Feng M, et al. Microgravity inhibits resting T cell immunity in an exposure time-dependent manner[J]. Int J Med Sci, 2014, 11(1):87-96.
doi: 10.7150/ijms.7651 |
| [8] |
El-Shafey EM, El-Nagar GF, El-Bendary AS, et al. Serum soluble interleukin-2 receptor alpha in systemic lupus erythematosus[J]. Iran J Kidney Dis, 2008, 2(2):80-85.
pmid: 19377213 |
| [9] | Hochberg MC. Updating the American College of Rheumatology revised criteria for the classification of systemic lupus erythematosus[J]. Arthritis Rheum, 1997, 40(9):1725. |
| [10] |
Gladman DD, Ibañez D, Urowitz MB. Systemic lupus erythematosus disease activity index 2000[J]. J Rheumatol, 2002, 29(2):288-291.
pmid: 11838846 |
| [11] |
Romero-Diaz J, Isenberg D, Ramsey-Goldman R. Measures of adult systemic lupus erythematosus: Updated version of British Isles Lupus Assessment Group (BILAG 2004), European Consensus Lupus Activity Measurements (ECLAM), Systemic Lupus Activity Measure, Revised (SLAM-R), Systemic Lupus Activity Questionnaire for Population Studies (SLAQ), Systemic Lupus Erythematosus Disease Activity Index 2000 (SLEDAI-2K), and Systemic Lupus International Collaborating Clinics/American College of Rheumatology Damage Index (SDI)[J]. Arthritis Care Res (Hoboken), 2011, 63(Suppl 11):S37-S46.
doi: 10.1002/acr.v63.11s |
| [12] |
Russell SE, Moore AC, Fallon PG, et al. Soluble IL-2Rα (sCD25) exacerbates autoimmunity and enhances the development of Th17 responses in mice[J]. PLoS One, 2012, 7(10):e47748.
doi: 10.1371/journal.pone.0047748 |
| [13] | Spolski R, Li P, Leonard WJ. Biology and regulation of IL-2: From molecular mechanisms to human therapy[J]. Nat Rev Immunol, 2018, 18(10):648-659. |
| [14] | Mizui M, Tsokos GC. Targeting regulatory T cells to treat patients with systemic lupus erythematosus[J]. Front Immunol, 2018(9):786. |
| [15] |
Laut J, Senitzer D, Petrucci R, et al. Soluble interleukin-2 receptor levels in lupus nephritis[J]. Clin Nephrol, 1992, 38(4):179-184.
pmid: 1424303 |
| [1] | 汤晓菲, 丁秋玲, 张阳, 王育梅, 田美伊, 孙卓, 王荣荣, 陈妍, 刘坚. 阿伐曲泊帕治疗难治性系统性红斑狼疮相关血小板减少1例[J]. 北京大学学报(医学版), 2026, 58(2): 405-409. |
| [2] | 李欣艺, 赵金霞, 穆荣. 阿司匹林对系统性红斑狼疮妊娠者结局的影响[J]. 北京大学学报(医学版), 2025, 57(6): 1074-1080. |
| [3] | 王晓林, 郭邵逸, 陈大召, 温锡杰, 华勇, 张亮, 张秦. 全髋关节置换术治疗系统性红斑狼疮继发股骨头缺血性坏死的随访研究[J]. 北京大学学报(医学版), 2025, 57(6): 1081-1088. |
| [4] | 卫春, 杨月, 赵新菊, 刘栩, 贾园. 系统性红斑狼疮合并自身免疫性郎飞结病1例[J]. 北京大学学报(医学版), 2025, 57(6): 1174-1179. |
| [5] | 梁景原, 张霞, 姚海红. 误诊为系统性硬化症的POEMS综合征1例[J]. 北京大学学报(医学版), 2025, 57(6): 1184-1187. |
| [6] | 赵凯, 鲁芙爱, 王永福. 中枢神经系统感染模拟神经精神狼疮1例[J]. 北京大学学报(医学版), 2025, 57(6): 1188-1192. |
| [7] | 王小梦, 曾晓君, 李娟. 黎族与汉族系统性红斑狼疮患者的主要临床特征[J]. 北京大学学报(医学版), 2025, 57(6): 1213-1218. |
| [8] | 曹沛, 栾庆先. 牙周炎与全身系统性疾病的思考与探索[J]. 北京大学学报(医学版), 2025, 57(5): 852-858. |
| [9] | 朱丽秀, 陈仁利, 周素娟, 林烨, 汤一榕, 叶桢. 水通道蛋白5对干燥综合征大鼠TLR4/MyD88/NF-κB信号的影响[J]. 北京大学学报(医学版), 2025, 57(5): 875-883. |
| [10] | 袁显墩, 李照华, 徐丹, 李婷, 方丹, 穆荣. 丝氨酸蛋白酶23在系统性硬化病皮肤纤维化中的作用和机制[J]. 北京大学学报(医学版), 2025, 57(5): 903-910. |
| [11] | 王文琼, 侯玉珂, 李春, 张学武. 系统性红斑狼疮患者不良妊娠结局的预测因素[J]. 北京大学学报(医学版), 2025, 57(3): 599-603. |
| [12] | 霍梦月, 梅花, 张钰恒, 张艳波, 刘春丽. miR-34a在高氧诱导新生大鼠支气管肺发育不良模型中的表达及调控机制[J]. 北京大学学报(医学版), 2025, 57(2): 237-244. |
| [13] | 王红彦, 李鑫铭, 房柯池, 朱华群, 贾汝琳, 王晶. 系统性红斑狼疮疾病活动度相关特征分析及评估模型的构建[J]. 北京大学学报(医学版), 2024, 56(6): 1017-1022. |
| [14] | 陈丹丹, 李云, 卢情怡, 相晓红, 孙峰, 李英妮, 赵静, 王红彦, 李春. 育龄期系统性红斑狼疮患者卵巢功能的评价及其影响因素[J]. 北京大学学报(医学版), 2024, 56(6): 1023-1028. |
| [15] | 王莉, 高超, 任欢欢, 沈艳平, 黄晓玮, 姚鸿, 韩丹丹. 系统性红斑狼疮患者自我管理能力现状及相关因素分析[J]. 北京大学学报(医学版), 2024, 56(6): 1029-1035. |
|
||