北京大学学报(医学版) ›› 2026, Vol. 58 ›› Issue (2): 285-289. doi: 10.19723/j.issn.1671-167X.2026.02.010
干细胞功能调控在颅颌面组织再生修复中的研究进展
- 北京大学口腔医学院·口腔医院正畸科, 国家口腔医学中心, 国家口腔疾病临床医学研究中心, 口腔生物材料和数字诊疗装备国家工程研究中心, 口腔数字医学北京市重点实验室, 北京 100081
Progress in regulating stem cell functions for repair and regeneration of craniomaxillofacial tissues
Han ZHANG, Fujia YANG, Ruili YANG*( )
)
- Department of Orthodontics, Peking University School and Hospital of Stomatology & National Center for Stomatology & National Clinical Research Center for Oral Diseases & National Engineering Research Center of Oral Biomaterials and Digi-tal Medical Devices & Beijing Key Laboratory of Digital Stomatology, Beijing 100181, China
摘要:
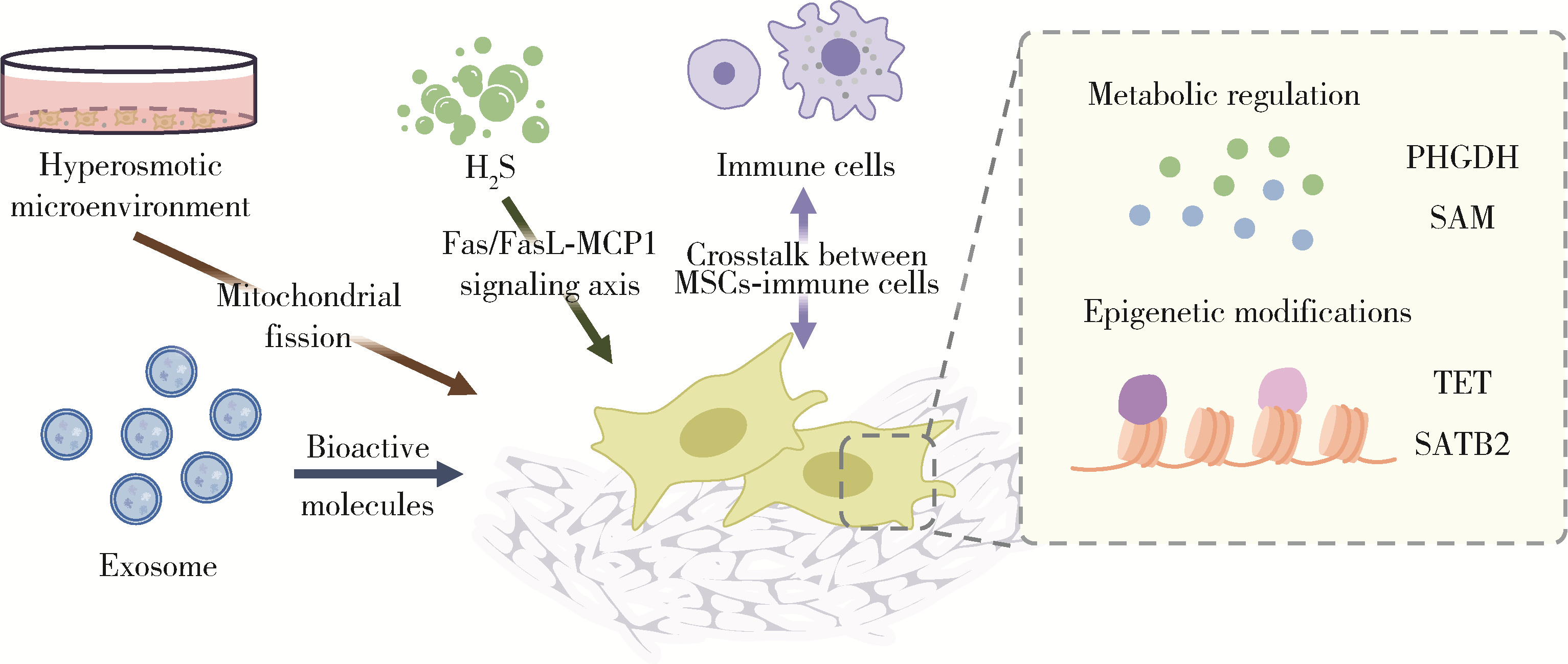
颅颌面组织再生修复是口腔与再生医学领域的关键挑战。间充质基质/干细胞(mesenchymal stromal/stem cells, MSCs)作为核心效应细胞, 其功能受代谢、表观遗传与免疫网络的多维精密调控。本文系统综述了MSCs在颅颌面组织再生中的功能调控机制, 重点探讨了力学刺激、代谢重编程、H2S气体信号以及表观遗传修饰对MSCs干性维持、定向分化及免疫调节功能的影响。同时, 阐述了MSCs通过外泌体等载体与免疫细胞相互作用, 共同调控骨稳态与再生进程的双向机制。文末展望了基于MSCs及其工程化外泌体的靶向治疗策略所面临的挑战与临床转化前景, 旨在为颅颌面缺损的再生修复提供新的见解。
中图分类号:
- R782
| 1 |
doi: 10.1016/j.scib.2025.03.011 |
| 2 |
doi: 10.1186/s13287-018-0965-3 |
| 3 |
doi: 10.1016/j.stemcr.2022.06.006 |
| 4 |
doi: 10.1186/s13287-018-0804-6 |
| 5 |
doi: 10.1177/0022034520958374 |
| 6 |
doi: 10.1007/s12274-024-6758-3 |
| 7 |
doi: 10.1038/s41420-018-0071-4 |
| 8 |
doi: 10.1038/s41467-018-04464-6 |
| 9 |
doi: 10.1038/s41419-019-2025-z |
| 10 |
doi: 10.1186/s13287-021-02660-8 |
| 11 |
doi: 10.1038/s41419-025-07577-1 |
| 12 |
|
| 13 |
doi: 10.1038/s41419-020-02788-0 |
| 14 |
doi: 10.1177/0022034520901710 |
| 15 |
doi: 10.1126/sciadv.adq6700 |
| 16 |
doi: 10.3389/fimmu.2022.860225 |
| [1] | 李峥, 吕珑薇, 张晓, 夏丹丹, 张萍, 刘云松, 周永胜. 基于干细胞和生物材料调控的口腔颅颌面骨再生的研究进展[J]. 北京大学学报(医学版), 2026, 58(2): 272-277. |
| [2] | 王昕莹, 程雪原, 张孟钧, 李菲, 段晋瑜, 乔静. 浓缩生长因子联合引导性组织再生术治疗下颌磨牙根分叉病变的疗效[J]. 北京大学学报(医学版), 2026, 58(2): 372-379. |
| [3] | 曾立婷, 程凯远, 刘中宁, 李健, 杨静文, 姜婷. miR-488-5p促进大鼠骨髓间充质干细胞成神经、成骨分化及神经化骨再生[J]. 北京大学学报(医学版), 2026, 58(1): 10-21. |
| [4] | 欧乾民, 李政诗, 牛露菡, 任倩慧, 刘欣雨, 毛学理, 施松涛. 口腔干细胞的生物学特性与转化研究[J]. 北京大学学报(医学版), 2025, 57(5): 827-835. |
| [5] | 王宇蓝, 曾浩, 张玉峰. 口腔种植中血浆基质的临床转化现状与前沿探索[J]. 北京大学学报(医学版), 2025, 57(5): 836-840. |
| [6] | 马保金, 李建华, 桑元华, 于洋, 仇吉川, 邵金龙, 李凯, 刘世岳, 杜密, 商玲玲, 葛少华. 基于微环境和干细胞调控的牙周组织再生关键技术的建立与应用[J]. 北京大学学报(医学版), 2025, 57(5): 841-846. |
| [7] | 盛春辉, 张晓, 吕珑薇, 周永胜. 人脂肪间充质干细胞外泌体对去势小鼠骨质疏松的预防[J]. 北京大学学报(医学版), 2025, 57(2): 217-226. |
| [8] | 李梦迪, 雷蕾, 刘中宁, 李健, 姜婷. siRNA沉默NLK基因促进神经化组织工程骨再生[J]. 北京大学学报(医学版), 2025, 57(2): 227-236. |
| [9] | 王昕莹, 程雪原, 张勇, 李菲, 段晋瑜, 乔静. 浓缩生长因子与自固化磷酸钙人工骨联合治疗牙周骨下袋缺损的疗效:临床和影像学评价[J]. 北京大学学报(医学版), 2025, 57(1): 42-50. |
| [10] | 帅婷, 郭艳艳, 林春平, 侯晓玫, 金婵媛. 敲减NPTX1促进人骨髓间充质干细胞成骨分化[J]. 北京大学学报(医学版), 2025, 57(1): 7-12. |
| [11] | 武志慧, 胡明智, 赵巧英, 吕凤凤, 张晶莹, 张伟, 王永福, 孙晓林, 王慧. miR-125b-5p修饰脐带间充质干细胞对系统性红斑狼疮的免疫调控机制[J]. 北京大学学报(医学版), 2024, 56(5): 860-867. |
| [12] | 陈晓颖,张一,李雨柯,唐琳,刘玉华. 不同种类聚合物对猪小肠黏膜下层支架仿生矿化的影响[J]. 北京大学学报(医学版), 2024, 56(1): 17-24. |
| [13] | 赵菡,卫彦,张学慧,杨小平,蔡晴,宁成云,徐明明,刘雯雯,黄颖,何颖,郭亚茹,江圣杰,白云洋,吴宇佳,郭雨思,郑晓娜,李文静,邓旭亮. 口腔硬组织修复材料仿生设计制备和临床转化[J]. 北京大学学报(医学版), 2024, 56(1): 4-8. |
| [14] | 潘媛,顾航,肖涵,赵笠君,汤祎熳,葛雯姝. 泛素特异性蛋白酶42调节人脂肪干细胞成骨向分化[J]. 北京大学学报(医学版), 2024, 56(1): 9-16. |
| [15] | 刘晓强,周寅. 牙种植同期植骨术围术期高血压的相关危险因素[J]. 北京大学学报(医学版), 2024, 56(1): 93-98. |
|
||