北京大学学报(医学版) ›› 2026, Vol. 58 ›› Issue (2): 342-350. doi: 10.19723/j.issn.1671-167X.2026.02.019
原发性胃淋巴瘤的内镜特征分析及疗效预测
魏竞尧1, 叶菊香2, 周美玲1, 付伟伟1, 刘鑫1, 翟康乐1, 石岩岩3, 丁士刚1,*( ), 张静1,*(
), 张静1,*( )
)
- 1. 北京大学第三医院消化科, 北京 100191
2. 北京大学第三医院病理科, 北京 100191
3. 北京大学第三医院临床流行病学研究中心, 北京 100191
Endoscopic characteristics of primary gastric lymphoma and prediction of treatment response
Jingyao WEI1, Juxiang YE2, Meiling ZHOU1, Weiwei FU1, Xin LIU1, Kangle ZHAI1, Yanyan SHI3, Shigang DING1,*( ), Jing ZHANG1,*(
), Jing ZHANG1,*( )
)
- 1. Department of Gastroenterology, Peking University Third Hospital, Beijing 100191, China
2. Department of Pathology, Peking University Third Hospital, Beijing 100191, China
3. Clinical Epidemiology Research Center, Peking University Third Hospital, Beijing 100191, China
摘要:
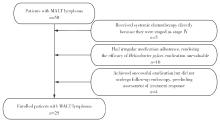
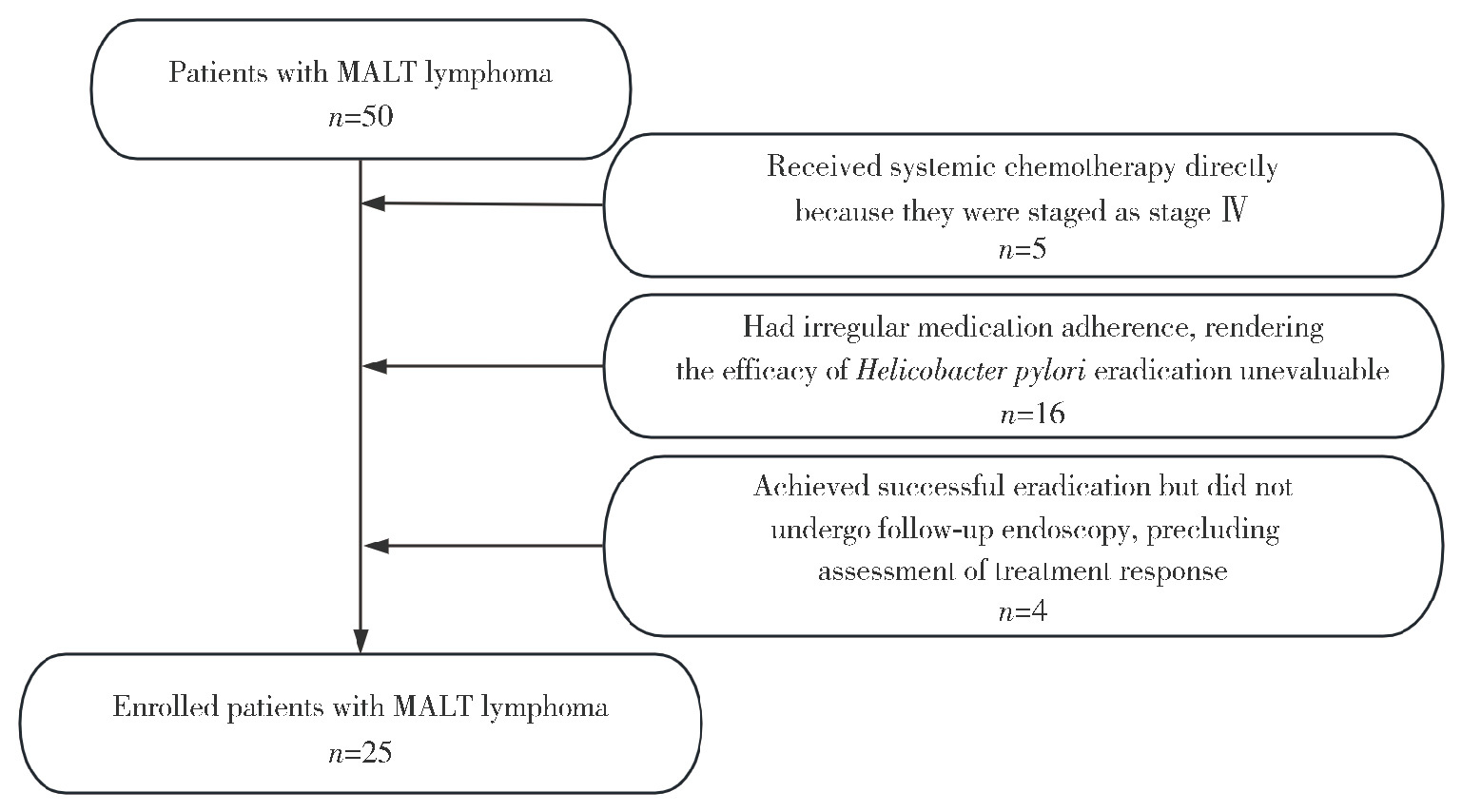
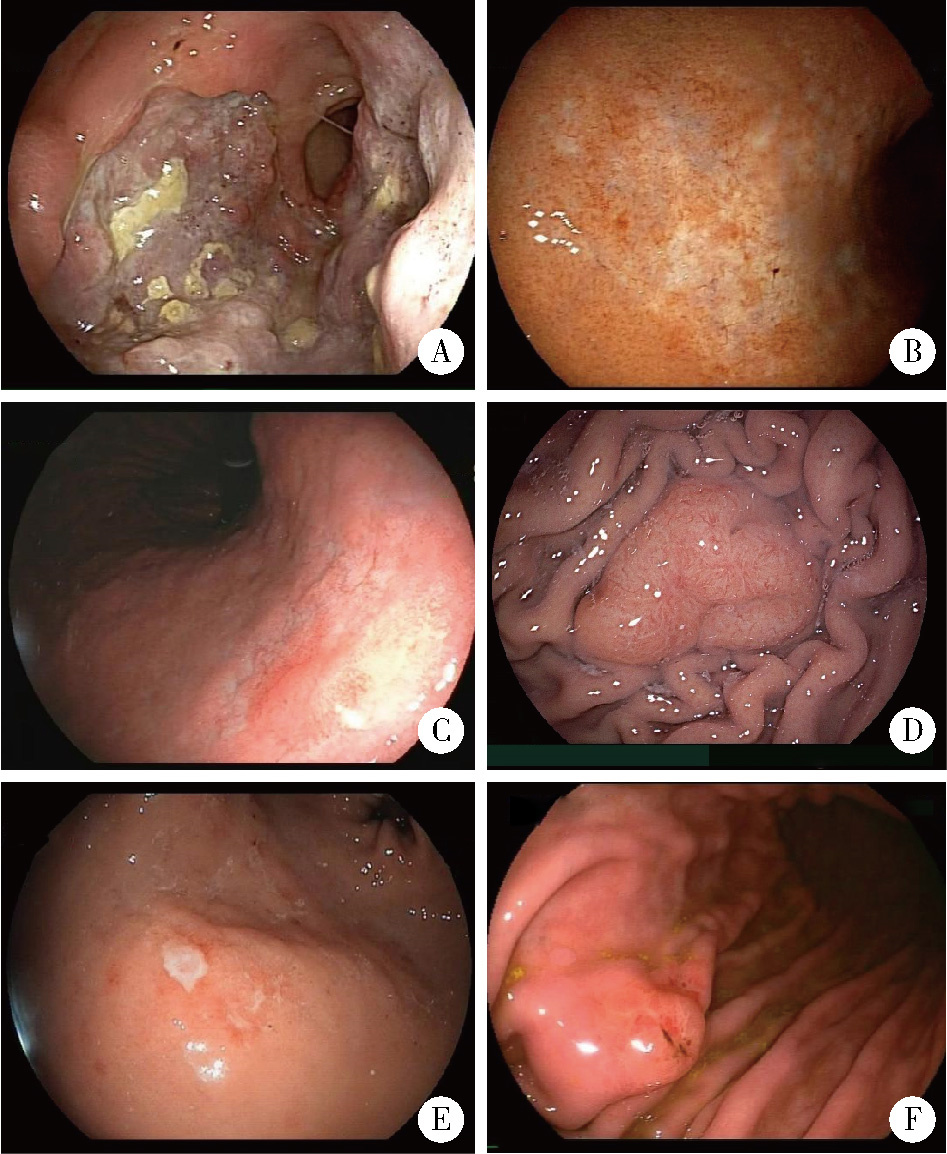
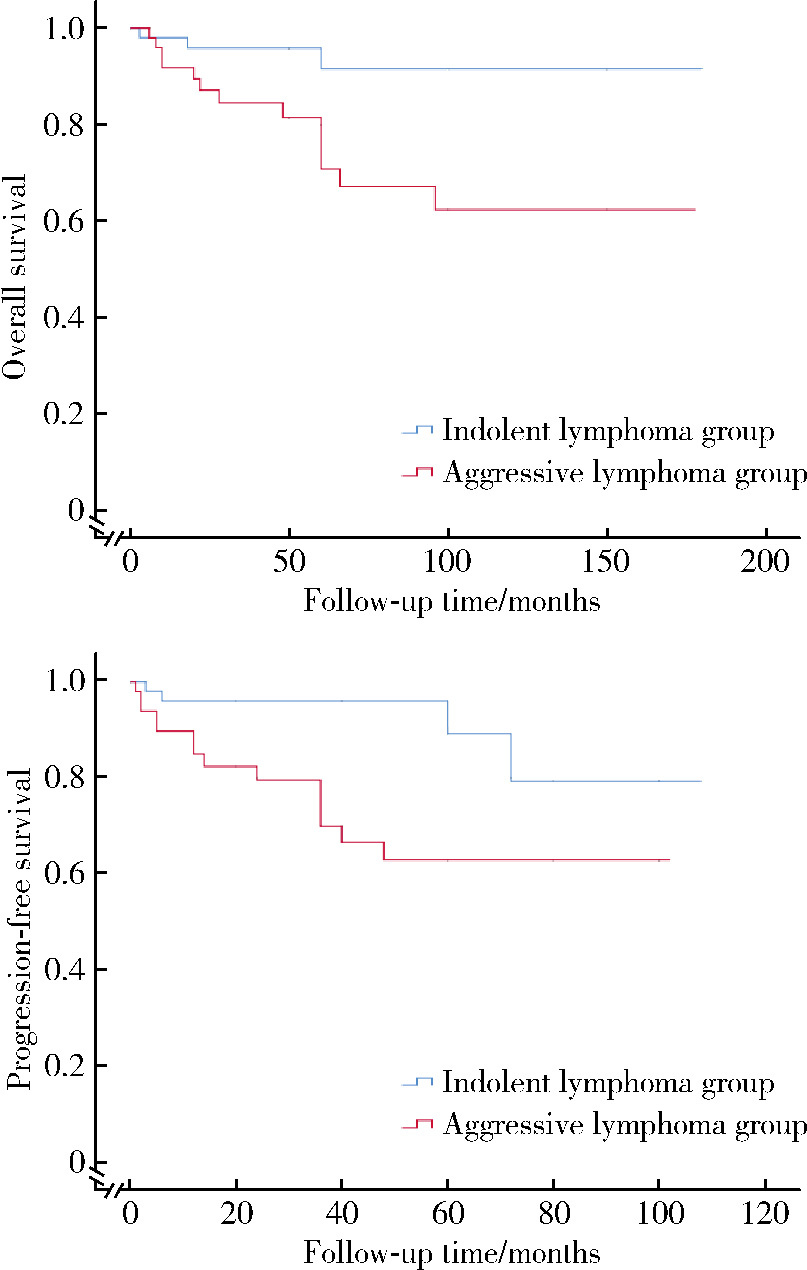
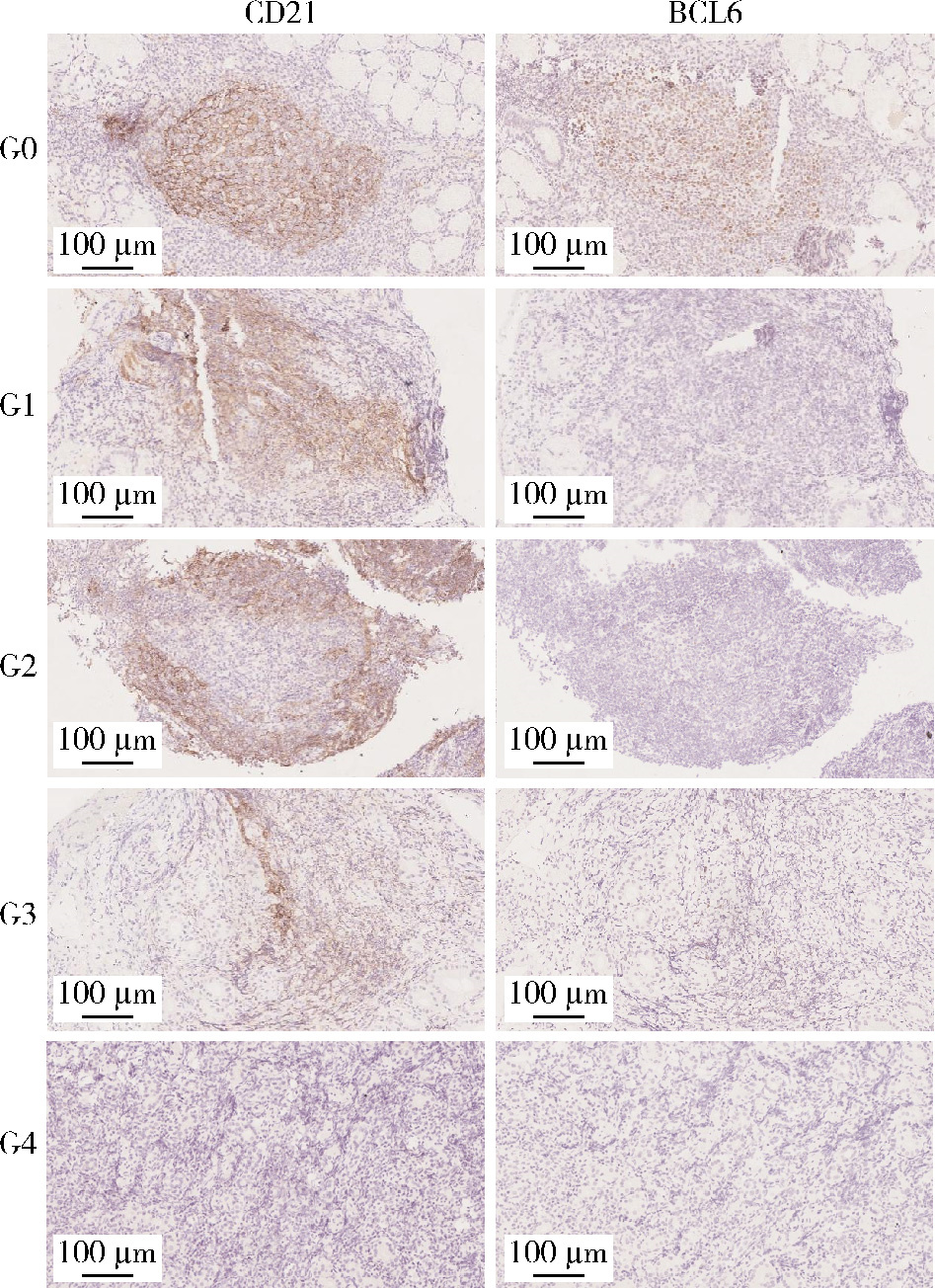
目的: 系统总结北京大学第三医院原发性胃淋巴瘤(primary gastric lymphoma, PGL)患者的内镜特征,探索基于补体受体2/B细胞淋巴瘤-6蛋白(complement receptor type 2/B-cell lymphoma 6 protein, CD21/BCL6)的淋巴滤泡破坏程度分级在胃黏膜相关淋巴组织(mucosa associated lymphoid tissue, MALT)淋巴瘤根除幽门螺杆菌(Helicobacter pylori, Hp)疗效中的预测价值。方法: 连续选择2010年1月至2025年1月北京大学第三医院确诊的100例PGL患者的病例资料进行回顾性分析,按病理类型分为惰性与侵袭性两组,比较其临床及内镜特征;采用Kaplan-Meier法绘制生存曲线并以Log-rank检验比较总生存率(overall survival, OS)和无进展生存率(progression free survival, PFS);另外选择同期根除Hp疗效明确的25例胃MALT淋巴瘤患者初诊活检标本行CD21/BCL6免疫组化染色并进行G0~G4分级,比较根除有效/无效亚组分级差异,并通过Logistic回归分析根除无效相关因素。结果: 共纳入100例PGL患者,平均年龄63.0(55.8, 71.0)岁,男性47例,女性53例;侵袭性组的B症状发生率高于惰性组(49.0% vs. 19.6%, P=0.004)。内镜方面,侵袭性组以溃疡型/混合型为主(P < 0.001),糜烂(98.0% vs. 49.0%, P < 0.001)、溃疡/白苔(96.0% vs. 37.3%, P < 0.001)、病灶“质脆”(47.0% vs. 11.9%, P < 0.001)、出血倾向(P=0.008)、胃腔狭窄(38.8% vs. 0, P < 0.001)及蠕动差(49.0% vs. 9.8%, P < 0.001)更常见;生存分析显示侵袭性组OS与PFS均劣于惰性组(OS: P=0.009;PFS: P=0.003)。在25例胃MALT淋巴瘤亚队列中,无效组Hp阴性比例更高(P=0.049),且淋巴滤泡破坏程度分级显著高于有效组(P=0.015);多因素Logistic回归提示破坏程度分级与根除无效独立相关(AOR=3.63,95%CI: 1.14~11.58,P=0.021),Hp感染状态未见独立相关(P=0.240)。结论: PGL内镜表现具有显著异质性,溃疡型/混合型、病灶脆性、出血倾向、腔狭窄及蠕动差更提示侵袭性淋巴瘤,并且与较差生存相关;在胃MALT淋巴瘤中,初诊活检基于CD21/BCL6的淋巴滤泡破坏程度分级可用于早期识别根除Hp无效高风险人群,为分层治疗与随访决策提供依据。
中图分类号:
- R735.2
| 1 |
doi: 10.1097/MEG.0000000000001651 |
| 2 |
National Comprehensive Cancer Network (NCCN). NCCN Guidelines Navigator: B-cell lymphomas. Version 3.2025[EB/OL]. [2025-10-01]. https://www.nccn.org/guidelines/guidelines-detail?category=1&id=1480.
|
| 3 |
doi: 10.1016/j.annonc.2025.07.014 |
| 4 |
doi: 10.3748/wjg.v17.i6.697 |
| 5 |
doi: 10.1093/oxfordjournals.annonc.a058869 |
| 6 |
doi: 10.1038/s41375-022-01620-2 |
| 7 |
doi: 10.1111/bjh.12078 |
| 8 |
doi: 10.1186/s12957-019-1659-8 |
| 9 |
Herlevic V, Reynolds SB, Morris JD. Gastric Lymphoma[M/OL]. In: StatPearls[Internet]. Treasure Island (FL): StatPearls Publishing, 2024[2025-10-01]. https://www.ncbi.nlm.nih.gov/books/NBK567799/.
|
| 10 |
doi: 10.3390/diagnostics14060607 |
| 11 |
doi: 10.3390/cancers15153811 |
| 12 |
doi: 10.1186/s12876-024-03476-5 |
| 13 |
doi: 10.1111/bjh.19064 |
| 14 |
doi: 10.1055/a-2685-2913 |
| 15 |
doi: 10.1111/bjh.19470 |
| 16 |
doi: 10.12998/wjcc.v10.i33.12447 |
| 17 |
doi: 10.1309/AJCPZQ1GXBBNG8OG |
| 18 |
doi: 10.1038/s41598-024-55663-9 |
| 19 |
doi: 10.1038/s41598-020-71792-3 |
| 20 |
doi: 10.1053/gast.2002.33047 |
| 21 |
doi: 10.1182/blood-2005-01-0004 |
| 22 |
doi: 10.3324/haematol.2019.228775 |
| 23 |
doi: 10.3390/cancers14020446 |
| 24 |
doi: 10.1007/s00277-023-05130-8 |
| 25 |
doi: 10.1016/j.ijrobp.2024.10.020 |
| [1] | 季加孚, 韦静涛, 季科, 步召德. 胃癌诊疗的瓶颈与破局:迈向精准化与智能化融合的新纪元[J]. 北京大学学报(医学版), 2026, 58(2): 231-238. |
| [2] | 步召德, 冯梦宇, 季科. 早期胃癌行前哨淋巴结导航手术的实践与思考[J]. 北京大学学报(医学版), 2026, 58(2): 239-243. |
| [3] | 罗必显, 刘洪铭, 谢伟勋, 龚渭华. 产甲胎蛋白胃癌的新临床特征和前沿科学问题[J]. 北京大学学报(医学版), 2026, 58(2): 257-265. |
| [4] | 刘友东, 吕亚军, 陈杰, 臧明德, 潘宏达, 刘晓文, 陆俊, 刘凤林. 全腹腔镜保留贲门胃底胃次全切除术治疗中上部胃癌的疗效及安全性[J]. 北京大学学报(医学版), 2026, 58(2): 301-306. |
| [5] | 李嘉临, 陈力侨, 唐家天, 吴艳, 王安强. 胃肝样腺癌转化治疗1例[J]. 北京大学学报(医学版), 2026, 58(2): 399-404. |
| [6] | 郭芷均, 俎明, 马超, 张贺军, 张静, 丁士刚. 同时性多发早期胃癌漏诊病灶的临床病理特征[J]. 北京大学学报(医学版), 2026, 58(2): 410-415. |
| [7] | 李斌, 梁寒. 机器人胃癌根治术:研究进展与实践挑战[J]. 北京大学学报(医学版), 2026, 58(2): 416-422. |
| [8] | 侯卫华, 宋书杰, 石中月, 刘露, 金木兰. 食管胃结合部具有显著空泡状核特征的神经内分泌癌1例[J]. 北京大学学报(医学版), 2025, 57(5): 1005-1009. |
| [9] | 李浙民, 季加孚, 李国新, 李子禹, 步召德, 高翔宇, 董迪, 唐磊, 邢晓芳, 贾淑芹, 郭婷, 张连海, 陕飞, 季鑫, 王安强. 胃癌精准诊疗技术的创建与推广[J]. 北京大学学报(医学版), 2025, 57(5): 864-867. |
| [10] | 侯卫华,宋书杰,石中月,金木兰. 幽门螺杆菌阴性早期胃癌的临床病理特征[J]. 北京大学学报(医学版), 2023, 55(2): 292-298. |
| [11] | 刘菊梅,梁丽,张继新,戎龙,张梓怡,吴悠,赵旭东,李挺. 411例早期胃癌及癌前病变内镜黏膜下剥离术标本的病理学评估[J]. 北京大学学报(医学版), 2023, 55(2): 299-307. |
| [12] | 牛占岳,薛艳,张静,张贺军,丁士刚. 胃腺瘤性息肉的内镜和病理特点及癌变的危险因素分析[J]. 北京大学学报(医学版), 2021, 53(6): 1122-1127. |
| [13] | 武颖超,蔡云龙,戎龙,张继新,刘金,汪欣. 早期胃癌淋巴结转移规律及内镜黏膜下剥离术治疗早期胃癌的疗效评价[J]. 北京大学学报(医学版), 2020, 52(6): 1093-1097. |
| [14] | 杨阳,刘毅强,王晓红,季科,李忠武,白健,杨爱蓉,胡颖,韩海勃,李子禹,步召德,吴晓江,张连海,季加孚. 单中心大样本Epstein-Barr病毒相关性胃癌亚型的临床病理及分子特征分析[J]. 北京大学学报(医学版), 2019, 51(3): 451-458. |
| [15] | 刘鑫,张静,王晔,张贺军,丁士刚,周丽雅. 早期胃癌白光内镜下特征分析[J]. 北京大学学报(医学版), 2019, 51(2): 302-306. |
|
||